|

CLICK ON weeks 0 - 40 and follow along every 2 weeks of fetal development
|
||||||||||||||||||||||||||||
|
Developmental biology - Low Birthweight Low birthweight linked to placental protein Although the cause remains poorly understood, most physicians blame low birth rate on a malfunction in the placenta that prevents the mother's bloodstream from delivering oxygen and nutrients to her fetus. In studying the human placenta, researchers looked at gene expression: the process by which a gene's DNA sequence is converted into proteins within cells. They compared the placentas of women who gave birth to healthy babies to the placentas of women who delivered low-birthweight infants, and found significantly higher levels of humanin in the latter group. Humanin protein protects the body against oxidative stress, which hampers the body's ability to defend itself against toxic free radicals (a type of particle linked to disease). This finding may help researchers unravel the reasons why low birthweight babies face a higher risk of obesity, high blood pressure, diabetes and heart disease as adults. Abstract Background: Intrauterine growth restriction (IUGR) results from a lack of nutrients transferred to the developing fetus, particularly oxygen and glucose. Increased expression of the cytoprotective mitochondrial peptide, humanin (HN), and the glucose transporter 8, GLUT8, has been reported under conditions of hypoxic stress. However, the presence and cellular localization of HN and GLUT8 in IUGR-related placental pathology remain unexplored. Thus, we undertook this study to investigate placental expression of HN and GLUT8 in IUGR-affected versus normal pregnancies. Results: We found 1) increased HN expression in human IUGR-affected pregnancies on the maternal aspect of the placenta (extravillous trophoblastic (EVT) cytoplasm) compared to control (i.e. appropriate for gestational age) pregnancies, and a concomitant increase in GLUT8 expression in the same compartment, 2) HN and GLUT8 showed a protein-protein interaction by co-immunoprecipitation, 3) elevated HN and GLUT8 levels in vitro under simulated hypoxia in human EVT cells, HTR8/SVneo, and 4) increased HN expression but attenuated GLUT8 expression in vitro under serum deprivation in HTR8/SVneo cells. Conclusion: There was elevated HN expression with cytoplasmic localization to EVTs on the maternal aspect of the human placenta affected by IUGR, also associated with increased GLUT8 expression. We found that while hypoxia increased both HN and GLUT8, serum deprivation increased HN expression alone. Also, a protein-protein interaction between HN and GLUT8 suggests that their interaction may fulfill a biologic role that requires interdependency. Future investigations delineating molecular interactions between these proteins are required to fully uncover their role in IUGR-affected pregnancies. Authors: Janzen C1, Lei MYY1, Jeong ISD2,3, Ganguly A3,4, Sullivan P5, Paharkova V2,3, Capodanno G2,3, Nakamura H2,3, Perry A1, Shin BC3,4, Lee KW2,3, Devaskar SU3,4. Funding: This work was supported by the Health Innovation Challenge Fund (grant HICF-1009-003), a parallel funding partnership between Wellcome and the UK Department of Health, the Wellcome Sanger Institute (grant WT098051), and the MRC Human Genetics Unit program. Related websites: British Society for Genetic Medicine The British Society for Genetic Medicine (BSGM, http://www.bsgm.org.uk) is an independent umbrella organisation encompassing the Clinical Genetics Society, Association for Clinical Genomic Science, Association of Genetic Nurses and Counsellors and the Foetal and Cancer Genetics Groups, thereby representing the views of approximately 1800 professionals who work as doctors, genetic counsellors, scientists and researchers in supporting families and individuals with genetic or genomic disorders. These professionals work to predict, diagnose, prevent and treat these disorders and have contributed to major research discoveries. The Wellcome Sanger Institute The Wellcome Sanger Institute is one of the world's leading genome centres. Through its ability to conduct research at scale, it is able to engage in bold and long-term exploratory projects that are designed to influence and empower medical science globally. Institute research findings, generated through its own research programmes and through its leading role in international consortia, are being used to develop new diagnostics and treatments for human disease. To celebrate its 25th year in 2018, the Institute is sequencing 25 new genomes of species in the UK. Find out more at http://www.sanger.ac.uk or follow @sangerinstitute Wellcome exists to improve health for everyone by helping great ideas to thrive. We're a global charitable foundation, both politically and financially independent. We support scientists and researchers, take on big problems, fuel imaginations and spark debate. http://www.wellcome.ac.uk Return to top of page | Apr 2, 2018 Fetal Timeline Maternal Timeline News News Archive 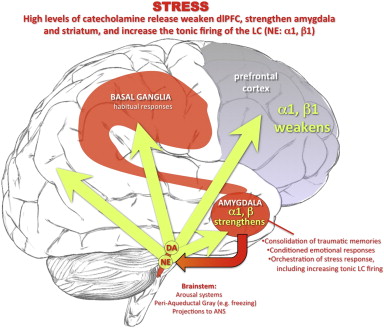 Under conditions of uncontrollable stress, high levels of catecholamine are released in the brain, weakening PFC function but strengthening the affective responses of the amygdala and habitual responses of the basal ganglia. The amygdala activates catecholamine under conditions of psychological stress, coordinating other aspects of the stress response (e.g. projections to the periaqueductal gray). Amygdala activation of the locus coeruleus (via CRF) increases tonic firing. High levels of NE release engage lower affinity alpha-1 and beta receptors, enhances amygdala and weakens PFC function, producing a vicious cycle that maintains primitive circuits in control of behavior.
|
||||||||||||||||||||||||||||

