|

CLICK ON weeks 0 - 40 and follow along every 2 weeks of fetal development
|
||||||||||||||||||||||||||||
|
Developmental Biology - CRISPR CRISPR Makes Stem Cells 'Invisible' Working with Professor Lewis Lanier PhD, study co-author and chair of UCSF's Department of Microbiology and Immunology, an expert in the signals that activate and inhibit NK cell activity - Schrepfer's team found that CD47, a cell surface protein that acts as a "do not eat me" signal against immune cells called macrophages, also has a strong inhibitory effect on NK cells. CD47 indeed proved to be the missing piece of the puzzle. When researchers transplanted their triple-engineered mouse stem cells into mismatched mice with normal immune systems, they observed no rejection. They then transplanted similarly engineered human stem cells into so-called humanized mice mice whose immune systems have been replaced with components of the human immune system to mimic human immunity and once again observed no rejection. Additionally, the researchers derived various types of human heart cells from these triple-engineered stem cells, which they again transplanted into humanized mice. The stem cell-derived cardiac cells were able to achieve long-term survival and even began forming rudimentary blood vessels and heart muscle, raising the possibility that triple-engineered stem cells may one day be used to repair failing hearts. "Our technique solves the problem of rejection of stem cells and stem cell-derived tissues, and represents a major advance for the stem cell therapy field. Our technique can benefit a wider range of people with production costs that are far lower than any individualized approach. We only need to manufacture our cells one time and we're left with a product that can be applied universally." Abstract Autologous induced pluripotent stem cells (iPSCs) constitute an unlimited cell source for patient-specific cell-based organ repair strategies. However, their generation and subsequent differentiation into specific cells or tissues entail cell line-specific manufacturing challenges and form a lengthy process that precludes acute treatment modalities. These shortcomings could be overcome by using prefabricated allogeneic cell or tissue products, but the vigorous immune response against histo-incompatible cells has prevented the successful implementation of this approach. Here we show that both mouse and human iPSCs lose their immunogenicity when major histocompatibility complex (MHC) class I and II genes are inactivated and CD47 is over-expressed. These hypoimmunogenic iPSCs retain their pluripotent stem cell potential and differentiation capacity. Endothelial cells, smooth muscle cells, and cardiomyocytes derived from hypoimmunogenic mouse or human iPSCs reliably evade immune rejection in fully MHC-mismatched allogeneic recipients and survive long-term without the use of immunosuppression. These findings suggest that hypoimmunogenic cell grafts can be engineered for universal transplantation. Authors Tobias Deuse, Xiaomeng Hu, Alessia Gravina, Dong Wang, Grigol Tediashvili, Chandrav De, William O. Thayer, Angela Wahl, J. Victor Garcia, Hermann Reichenspurner, Mark M. Davis, Lewis L. Lanier and Sonja Schrepfer. Conflicts: The authors declare no competing financial interests. Acknowledgements The authors thank C. Pahrmann for cell culture work, imaging experiments and overall assistance and L. Li for his assistance. The in vivo BLI experiments were performed at the UCSF Pre-clinical Therapeutics Core (A. Fries; with special thanks to B.C. Hann). Special thanks go to J. Wu (Stanford Cardiovascular Institute, Stanford University School of Medicine) for providing the miPSCs and the help of his laboratory with developing the protocol for hiPSC differentiation into cardiomyocytes. We thank J.-F. Garcia-Gomez (City of Hope, Duarte) for the HLA typing of humanized BLT mice. We also thank E. Maltepe and H. Pektas for providing the syncytiotrophoblast cells. D.W. was supported by the Max Kade Foundation. A.W. received funding from the National Institutes of Health (grant AI123010). J.V.G. received funding from the National Institutes of Health (AI111899 and MH108179). The cardiomyocyte research was partly made possible by a grant from the California Institute for Regenerative Medicine (Grant Number DISC1-09984). Research related to cardiomyocyte immunobiology reported in this publication was supported by National Heart, Lung, and Blood Institute of the National Institutes of Health under award number R01HL140236. L.L.L. is an American Cancer Society Professor funded by NIH AI068129 and in part by the Parker Institute for Cancer Immunotherapy. S.S. and T.D. received funding for the cardiomyocyte research from the California Institute for Regenerative Medicine (Grant Number DISC1-09984) and for the immunobiology work from the National Heart, Lung, and Blood Institute of the National Institutes of Health under award number R01HL140236. The contents of this publication are solely the responsibility of the authors and do not necessarily represent the official views of the NIH, CIRM and other agencies of the State of California. This work was supported by National Institutes of Health grants R01NS095884, EB003392, R01EB016657, R01CA185363, 1S10RR019003-01 and 1S10RR025488-01; National Multiple Sclerosis Society grant 5045A1, and National Science Foundation grant CMMI-1634888. About UCSF: UC San Francisco (UCSF) is a leading university dedicated to promoting health worldwide through advanced biomedical research, graduate-level education in the life sciences and health professions, and excellence in patient care. It includes top-ranked graduate schools of dentistry, medicine, nursing and pharmacy; a graduate division with nationally renowned programs in basic, biomedical, translational and population sciences; and a preeminent biomedical research enterprise. It also includes UCSF Health, which comprises three top-ranked hospitals - UCSF Medical Center and UCSF Benioff Children's Hospitals in San Francisco and Oakland - as well as Langley Porter Psychiatric Hospital and Clinics, UCSF Benioff Children's Physicians and the UCSF Faculty Practice. UCSF Health has affiliations with hospitals and health organizations throughout the Bay Area. UCSF faculty also provide all physician care at the public Zuckerberg San Francisco General Hospital and Trauma Center, and the SF VA Medical Center. The UCSF Fresno Medical Education Program is a major branch of the University of California, San Francisco's School of Medicine. Please visit http://www.ucsf.edu/news. Funding: Research was supported by grants from the Deutsche Forschungsgemeinschaft, the Fondation Leducq, the Max Kade Foundation, the California Institute for Regenerative Medicine, the National Institutes of Health and the Parker Institute for Cancer Immunotherapy. Return to top of page | Feb 20, 2019 Fetal Timeline Maternal Timeline News 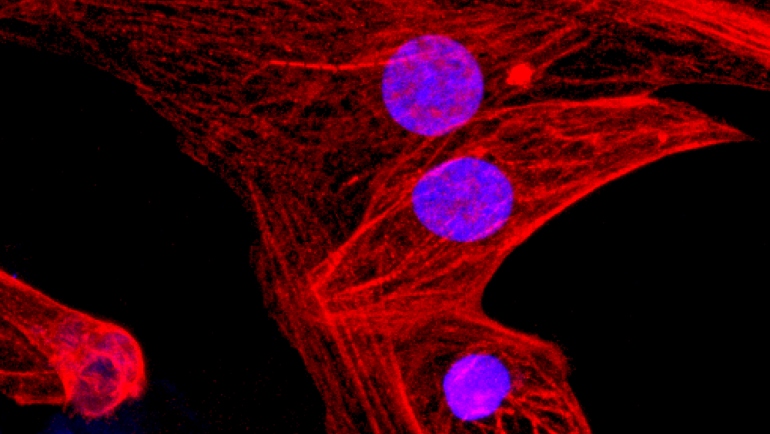 Human heart muscle cells derived from triple-engineered stem cells that are invisible to the immune system. RED is troponin, a protein that participates in cardiac muscle contraction. Cell nuclei are BLUE. Researchers hope cells like these will eventually be used to treat heart failure. Image Credit: Xiaomeng Hu.
|
||||||||||||||||||||||||||||

