|
|
Developmental Biology - Mitochondria
Eliminating Damaged Mitochondria
Treatment with an inhibitor of choline kinase prompts immune cells to clear away damaged mitochondria, and reduce and prevent inflammation...
Inflammation is a physiologic response. The body needs it to eliminate invasive organisms and foreign irritations. But in excess, it harms healthy cells and contributes to aging and chronic diseases. In order to regulate inflammation, immune cells use a molecular machine called NLRP3 inflammasome which is inactive in a healthy cell, but turns on when a cell's energy-generating organelles - mitochondria - become damaged by stress or exposure to the toxins produced by bacteria.
When the NLRP3 inflammasome gets stuck in the "on" position, it can also contribute to other chronic inflammatory conditions such as gout, osteoarthritis, fatty liver disease, Parkinson's and Alzheimer's diseases.
In a new mouse study, researchers at the University of California San Diego School of Medicine, discovered a unique approach that might help treat some of these chronic inflammatory diseases. Their approach forces cells to eliminate damaged mitochondria before they can activate NLRP3 inflammasome.
Published April 11, 2019 in Cell Metabolism, the study was led by senior author Michael Karin PhD, professor, pharmacology and pathology, UC San Diego School of Medicine. First author Elsa Sanchez-Lopez PhD, is the senior postdoctoral researcher in Karin's lab.
In a study published 2018 in Nature, Karin's team showed how damaged mitochondria activate the NLRP3 inflammasome.
In 2018, Karin's researchers found NLRP3 inflammasome is de-activated when mitochondria are removed by the cell's internal waste recycling process, called mitophagy.
Karin: "After that, we wondered if we could reduce harmful excess inflammation by intentionally inducing mitophagy, which would eliminate damaged mitochondria and should in turn pre-emptively inhibit NLRP3 inflammasome activation. But, we didn't have a good way to induce mitophagy."
While Sanchez-Lopez was studying how macrophages regulate their uptake of choline, a nutrient critical to metabolism, she came across something that can initiate mitophagy: an inhibitor of the enzyme choline kinase (called ChoK). With ChoK inhibited, choline is no longer incorporated into mitochondrial membranes. As a result, the cells perceive mitochondria as damaged, and cleared them away by mitophagy.
"By getting rid of damaged mitochondria using the enzyme choline kinase - ChoK inhibitors - we were finally able to inhibit NLRP3 inflammasome activation."
Michael Karin PhD, Distinguished Professor of Pharmacology and Pathology; the Ben and Wanda Hildyard Chair for Mitochondrial and Metabolic Diseases, University of California, San Diego School of Medicine, California, USA.
To test their new ability to control NLRP3 inflammasome in a living system, researchers tested on mice. They discovered treatment with ChoK inhibitors prevented acute inflammation caused by bacterial toxins and uric acid (accumulation of which triggers gout flares).
ChoK inhibitor also reversed chronic inflammation associated with the genetic disease "Muckle-Well Syndrome" caused by mutations in NLRP3 genes. The larger the spleen, the more inflammation. Muckle-Well mice spleens average twice as large as normal.
But spleen size normalized after ChoK inhibitor treatment, as evidenced by smaller spleens on the right compared to mock-treated mice (three larger spleens on the left).
Karin: "There are several diseases, including lupus and osteoarthritis, whose treatment will likely require dual inhibition of both IL-1ß and IL-18." NLRP3 inflammasome promotes inflammation as it triggers release of two very potent pro-inflammatory molecules - the cytokines: interleukin IL-1ß and IL-18.
According to Karin, there are existing drugs that block IL-1ß, but not IL-18. However, his team found ChoK inhibitors can reduce both.
Highlights
LPS stimulates choline uptake and phosphorylation via CTL1 induction
Impaired choline uptake or phosphorylation reduces mitochondrial ATP synthesis
Reduced ATP synthesis leads to AMPK activation and induction of mitophagy
By causing mitophagy, ChoKa inhibition prevents NLRP3-dependent inflammation
Summary
Choline is a vitamin-like nutrient that is taken up via specific transporters and metabolized by choline kinase, which converts it to phosphocholine needed for de novo synthesis of phosphatidylcholine (PC), the main phospholipid of cellular membranes. We found that Toll-like receptor (TLR) activation enhances choline uptake by macrophages and microglia through induction of the choline transporter CTL1. Inhibition of CTL1 expression or choline phosphorylation attenuated NLRP3 inflammasome activation and IL-1ß and IL-18 production in stimulated macrophages. Mechanistically, reduced choline uptake altered mitochondrial lipid profile, attenuated mitochondrial ATP synthesis, and activated the energy sensor AMP-activated protein kinase (AMPK). By potentiating mitochondrial recruitment of DRP1, AMPK stimulates mitophagy, which contributes to termination of NLRP3 inflammasome activation. Correspondingly, choline kinase inhibitors ameliorated acute and chronic models of IL-1ß-dependent inflammation.
Authors
Elsa Sanchez-Lopez, Zhenyu Zhong, Alexandra Stubelius,, Shannon R. Sweeney, Laela M. Booshehri, Laura Antonucci, Ru Liu-Bryan, Alessia Lodi, Robert Terkeltaub, Juan Carlos Lacal, Anne N. Murphy, Hal M. Hoffman, Stefano Tiziani, Monica Guma and Michael Karin.
Return to top of page
| |
|
Apr 16 2019 Fetal Timeline Maternal Timeline News
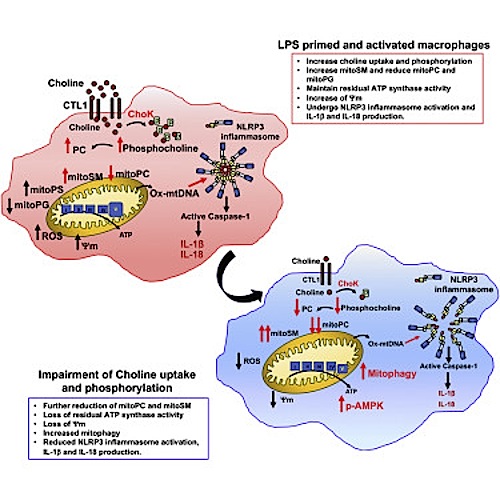 Choline is a vitamin-like nutrient which converts to phospho-choline needed for de novo synthesis of PhosphatidylCholine (PC), the main phospholipid of cell membranes. Activating Toll-like receptor (TLR) increases choline uptake altering mitochondrial lipids and activates AMPK. AMPK stimulates mitophagy, aiding the end of inflammation. CREDIT: Michael Karin, lead contqact.
|




