|
|
Developmental Biology - Heart Valve Defects
Fetal Cilia & Adult Heart Valves
Heart valve cell genetic variation creates a blueprint for mitral valve prolapse...
Mitral valve prolapse is a common congenital abnormality which can cause severe complications. After combining studies of mitral valve development in mice along with human genetic data, an international research collaboration including scientists, geneticists, cardiologists, echo-cardiographers and surgeons, has found adult mitral valve prolapse begins with abnormal cilia in the fetal heart.
The authors tracked mitral valve development in mice from fetus to adulthood, at the same time analyzing human patients suffering with the disease. One family in particular with an inherited form caused by a mutation in the DZIP1 gene which regulates ciliogenesis the building of cell antenna or primary cilia. Mice with this mutation also develop mitral valve prolapse. The research is published in Science Translational Medicine.
Previously there was no awareness that DZIP1 gene mutations affected adult heart valves function.
"These tiny structures are really important in establishing how the heart valve develops in utero," says Russell A. Norris PhD, who has studied heart valve disease for more than 20 years and is senior author on the article and associate professor of medicine in the Department of Regenerative Medicine and Cell Biology the Medical University of South Carolina (MUSC).
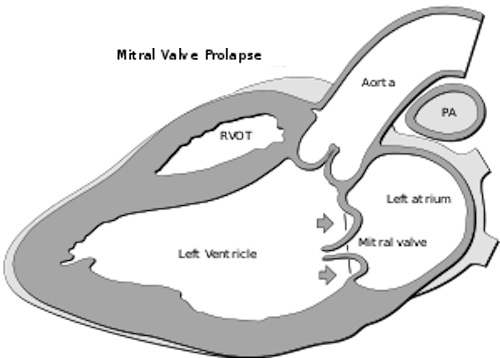
The mitral valve job is to keep blood flowing in one direction from the left atrium to the left ventricle of the heart. But, if it becomes enlarged it can't close tightly, allowing blood flow in both directions. Many of the one in forty Americans with mitral valve prolapse experience minimal symptoms. However, they can still experience damage to the heart muscle itself which can latter result in congestive heart failure, scarring of the heart tissue, and in serious cases, sudden cardiac death.
The research collaborative team initially assembled for a Leducq Foundation-funded project culminating in 2015 Nature and Nature Genetics articles announcing identification of genetic variants that cause mitral valve prolapse. Researchers who continued their collaboration, have since noticed each of the genes they previously identified can now be linked to a pathway involving primary cell cilia. This led them to test whether removing cilia in mice would cause something like mitral valve prolapse. "We have ways of removing cilia genetically in animal models," explains Norris. "We thought that if we took out the cilia in heart valve cells, then the mice should develop the same disease. And that turned out to be the case."
Next, the team looked at a multigenerational family with a history of mitral valve prolapse to see if there was additional evidence for a causative role for cilia in the disease. "Through our existing, long-term collaborations with geneticist Dr. Susan Slaugenhaupt, cardiologist Dr. David Milan and echocardiographer Dr. Robert Levine at Mass General Hospital, genetic analyses of this family were performed," says Norris. "Through many years of study we were able to identify a causative gene in this family, which ended up being a cilia gene."
To further support that finding, Norris developed a mouse model of mitral valve prolapse. Instead of knocking out whole genes, as others had done, Norris and his collaborators at MUSC used CRISPR CAS-9 technology to "knock in" the genetic mutation identified in the large family. Norris: "We knocked in the mutation and it fully recapitulated the phenotypic spectrum of the disease. impairing formation of primary cilia during development. These mice were quite beneficial as they confirmed that mitral valve prolapse could be caused by a defect in cilia formation during embryonic development."
Finally, the research team used an existing dataset from a previous genome-wide association study which looked at 300 known cilia genes, to see if there was an association between those variants and mitral valve prolapse. "We found that there was an increased prevalence of cilia gene variants in the mitral valve prolapse population," says Norris. "Whether it's relevant to ten, 40, or 70 percent of the broader population, we don't know yet."
Norris now hopes to use the unique mouse model the team has developed to better understand the temporal progression of the disease. "Mitral valve disease is most commonly seen when somebody in their fifties or sixties," explains Norris. "Yet, all of these genes that we've identified that cause mitral valve prolapse do not appear to be expressed in the adult. So how can a developmental error result in an adult pathology?"
The collaborative data indicates mitral valve prolapse may be present early on but clinically benign and not likely to present any symptoms. The disease can take decades to become clinically relevant representing a slow degeneration of valve tissue. Possibly contributing to degeneration are changes in heart geometry along with valve anatomy causing secondary defects.
"We think the geometry change, combined with altered biomechanics plus additional secondary responses that have yet to be discovered, result over time in the tissue being destroyed," says Norris. Norris will use his unique mouse model to test that theory. "We're the only group in the world that has this particular model," states Norris. "We want to use this model as our test bed to look at the mechanisms and timing by which the disease progresses."
Norris also hopes to begin to identify biomarkers of disease severity that could inform treatment. He will work with cardiothoracic surgeons in Leipzig, Germany and echocardiographers in Boston, who are collecting biopsy specimens from patients with mitral valve prolapse. He will compare those specimens with tissue biopsies from his animal model."Once we have the biopsies, we can then correlate changes in protein profiles in valve and heart tissues in humans with those in our animal models to look for markers of disease severity and/or progression," explains Norris. "Is there something predictive in the patients that will allow us to know that they are at a particular stage of fibrosis or heart failure or let us know what sort of outcomes they are likely to have?"
The findings of a developmental cause for some forms of the disease by Norris and other members of the collaborative team could signal the importance of early intervention and lead to rethinking of current treatment guidelines.
"If you have mitral valve prolapse, our findings suggest that you may not want to wait for intervention," says Norris. "There's a good chance that your heart is already being stressed and that changes in the heart muscle have already begun, which may be difficult to reverse. So earlier intervention is likely better. We're hopeful that, with the help of additional funding, some of our discoveries will help change guidelines for the timing of surgical intervention as well as lead to non-surgical therapies to treat the disease."
Abstract
Mitral valve prolapse (MVP) affects 1 in 40 people and is the most common indication for mitral valve surgery. MVP can cause arrhythmias, heart failure, and sudden cardiac death, and to date, the causes of this disease are poorly understood. We now demonstrate that defects in primary cilia genes and their regulated pathways can cause MVP in familial and sporadic nonsyndromic MVP cases. Our expression studies and genetic ablation experiments confirmed a role for primary cilia in regulating ECM deposition during cardiac development. Loss of primary cilia during development resulted in progressive myxomatous degeneration and profound mitral valve pathology in the adult setting. Analysis of a large family with inherited, autosomal dominant nonsyndromic MVP identified a deleterious missense mutation in a cilia gene, DZIP1. A mouse model harboring this variant confirmed the pathogenicity of this mutation and revealed impaired ciliogenesis during development, which progressed to adult myxomatous valve disease and functional MVP. Relevance of primary cilia in common forms of MVP was tested using pathway enrichment in a large population of patients with MVP and controls from previously generated genome-wide association studies (GWAS), which confirmed the involvement of primary cilia genes in MVP. Together, our studies establish a developmental basis for MVP through altered cilia-dependent regulation of ECM and suggest that defects in primary cilia genes can be causative to disease phenotype in some patients with MVP.
Authors
Katelynn A. Toomer, Mengyao Yu, Diana Fulmer, Lilong Guo, Kelsey S. Moore, Reece Moore, Kala D. Drayton, Janiece Glover, Neal Peterson, Sandra Ramos-Ortiz, Alex Drohan, Breiona J. Catching, Rebecca Stairley, Andy Wessels, Joshua H. Lipschutz, Francesca N. Delling, Xavier Jeunemaitre, Christian Dina, Ryan L. Collins, Harrison Brand, Michael E. Talkowski, Federica del Monte, Rupak Mukherjee, Alexander Awgulewitsch, Simon Body, Gary Hardiman, E. Starr Hazard, Willian A. da Silveira, Baolin Wang, Maire Leyne, Ronen Durst, Roger R. Markwald, Solena Le Scouarnec, Albert Hagege, Thierry Le Tourneau, Peter Kohl, Eva A. Rog-Zielinska, Patrick T. Ellinor, Robert A. Levine, David J. Milan, Jean-Jacques Schott, Nabila Bouatia-Naji, Susan A. Slaugenhaupt and Russell A. Norris.
Acknowledgement
Founded in 1824 in Charleston, MUSC is the oldest medical school in the South, as well as the state's only integrated, academic health sciences center with a unique charge to serve the state through education, research and patient care. Each year, MUSC educates and trains more than 3,000 students and 700 residents in six colleges: Dental Medicine, Graduate Studies, Health Professions, Medicine, Nursing and Pharmacy. The state's leader in obtaining biomedical research funds, in fiscal year 2018, MUSC set a new high, bringing in more than $276.5 million. For information on academic programs, visit http://musc.edu.
As the clinical health system of the Medical University of South Carolina, MUSC Health is dedicated to delivering the highest quality patient care available, while training generations of competent, compassionate health care providers to serve the people of South Carolina and beyond. Comprising some 1,600 beds, more than 100 outreach sites, the MUSC College of Medicine, the physicians' practice plan, and nearly 275 telehealth locations, MUSC Health owns and operates eight hospitals situated in Charleston, Chester, Florence, Lancaster and Marion counties. In 2018, for the fourth consecutive year, U.S. News & World Report named MUSC Health the number one hospital in South Carolina. To learn more about clinical patient services, visit http://muschealth.org.
MUSC and its affiliates have collective annual budgets of $3 billion. The more than 17,000 MUSC team members include world-class faculty, physicians, specialty providers and scientists who deliver groundbreaking education, research, technology and patient care.
Return to top of page
| |
|
Jun 5 2019 Fetal Timeline Maternal Timeline News
 Genetic variations in primary cilia genes cause defects during valve morphogenesis in fetus and can progress to mitral valve prolapse in the adult. CREDIT K. Toomer & R. Norris, Medical University of South Carolina
|