|
|
Developmental Biology - Gene Error
How Cells Defend Against Genetic Error
Researchers discover mechanisms behind gene damage in proteins...
A cell contains transcripts of our genetic material, which migrates from the cell nucleus into the cytoplasm of the cell. This movement protects genetically transcribed material from being recruited or 'used up' by the "spliceosomes". If this protection doesn't occur, the entire cell is in danger as cancer and neurodegenerative diseases may develop from poorly constructed proteins.
Researchers at the University of Göttingen and the University Medicine Centre Göttingen demonstrated these underlying cellular mechanisms in their observations published in Cell Reports.
Human cells are made up of:
cell nucleus with DNA blueprints for the organism
cytoplasm where proteins are built.
In the nucleus, DNA is re-written into messenger RNA creating instructions for producing proteins. Separated from its original transcription, proteins can now be made in the cytoplasm.
Separation of pre-messenger RNA is important as it is not immediately usable. It still contains areas that must be excised before entering the cytoplasm. If not removed before entering the cytoplasm, shortened or dysfunctional proteins get produced which are dangerous to the cell.
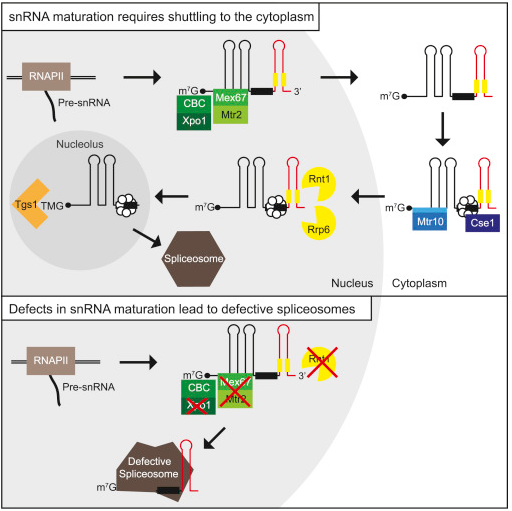
The molecular machinery that cuts these areas, non-coding regions or introns, out of the messenger RNA are the spliceosomes. They contain proteins and another type of DNA transcript snRNA. snRNA or small nuclear RNA is not translated into proteins like messenger RNA, but together with proteins forms the spliceosome molecular machinery.
In human cells, the snRNA in spliceosomes can also enter into the cytoplasm. In other organisms, such as baker's yeast often used as a model organism in research scientists had thought spliceosome snRNA never left the cell nucleus. Why the evolutionary development of exporting snRNA before incorporation into spliceosomes of human cells was a mystery.
"Our experiments show that in fact yeast spliceosome snRNA also migrates into the cytoplasm."
Heike Krebber PhD, Professor, Head, Department of Molecular Genetics, Institute for Microbiology and Genetics, University of Göttingen, Sweeden.
This second aspect of the research answered the question through genetic experiments, as the team manipulated yeast so that precursor snRNA no longer changed while in the cytoplasm to observe that: "The spliceosomes attempt to work with snRNA precursors, or unfinished snRNA, means it cannot function as it's supposed to," explains Krebber.
"The reason healthy cells must first send precursors of messenger RNA out of the cell nucleus immediately after its production, is to prevent messenger RNA from being used by the developing spliceosomes. This is important in order to identify any underlying cause and development of diseases.
Heike Krebber PhD
Highlights
All yeast snRNAs, including U6, shuttle into the cytoplasm
Export is mediated by Mex67 and Xpo1, and re-import requires Mtr10 and Cse1
snRNA export prevents an incorporation of immature snRNAs into spliceosomes
Spliceosomal assembly with immature snRNAs results in genome-wide splicing defects
Summary
Removal of introns from pre-mRNAs is an essential step in eukaryotic gene expression, mediated by spliceosomes that contain snRNAs as key components. Although snRNAs are transcribed in the nucleus and function in the same compartment, all - except U6 - shuttle to the cytoplasm. Surprisingly, the physiological relevance for shuttling is unclear, in particular because the snRNAs in Saccharomyces cerevisiae were reported to remain nuclear. Here, we show that all yeast pre-snRNAs including U6 undergo a stepwise maturation process after nuclear export by Mex67 and Xpo1. Sm- and Lsm-ring attachment occurs in the cytoplasm and is important for the snRNA re-import, mediated by Cse1 and Mtr10. Finally, nuclear pre-snRNA cleavage and trimethylation of the 5?-cap finalizes shuttling. Importantly, preventing pre-snRNAs from being exported or processed results in faulty spliceosome assembly and subsequent genome-wide splicing defects. Thus, pre-snRNA export is obligatory for functional splicing and resembles an essential evolutionarily conserved quality assurance step.
Authors
Daniel Becker, Anna Greta Hirsch, Lysann Bender, Thomas Lingner, Gabriela Salinas and Heike Krebber.
Acknowledgments
The authors thank H. Bastians and G. Zander for discussion and comments on the manuscript. We are also very thankful to G. Matera for his thoughtful comments and suggestions. We are grateful to J. Beggs, R. Bordonné, P. Fabrizio, R. Ficner, R. Lill, R. Lührmann, P.A. Silver, E. Hurt, B. Seraphin, K. Weis, and S. Wente for providing plasmids, strains, or antibodies. This work was funded by grants of the GGNB to D.B. and the Deutsche Forschungsgemeinschaft (DFG) and the SFB860 awarded to H.K.
Return to top of page
| |
|
Jun 20 2019 Fetal Timeline Maternal Timeline News
 cells in the cytoplasm 5271.png) Detection of snRNA in the cytoplasm of Normal or 'Wild Type' cells (LEFT). 'Capture' of snRNAs in cytoplasm of Genetically Modified cells (RIGHT). CREDIT Heike Krebber.
|

 cells in the cytoplasm 5271.png)


