|
|
Developmental Biology - Stem Cell Specialization
Size Matters!
New data reveals Cell size sparks genome awakening in embryos...
There is a transition period in early development, when an embryo switches from control by its mother's genes to control by its own genes. Now, researchers at the Perelman School of Medicine at the University of Pennsylvania, reveal that an embryo activates its genes (its genome) not all at once, but in a pattern based on the size of its cells.
Using the African clawed frog (Xenopus laevis) as their experimental animal, scientists recorded how cell size governs when genes turn on in a zygote (the name given newly fused egg and sperm). Cells of the zygote must achieve a threshold size before large-scale transcription of their own and not their mother's, proteins initiates. By generating miniature embryos, the team found that changes in cell size is what controls activation of a zygote's singularly unique genome.
Their research results are published as the cover story in Developmental Cell.
Maternal RNA and proteins regulate the cell cycle in an early embryo just undergoing cell division. The genome of the zygote the fertilized egg are initially in sleep mode. Yet at some point, zygote nuclei wake up and their newly formed genome takes over biochemical control for all subsequent development. But how an embryo recognizes when to undergo this transition was unknown.
"How an embryo 'hands over' control of development from mother to zygote is a fundamental question in developmental biology. Previously it wasn't appreciated how different regions of a vertebrate embryo can undergo genome activation at different times, or how directly cell size regulates wakens a zygote's genome."
Matthew C. Good PhD, Assistant Professor, Cell and Developmental Biology and Bioengineering, the Raymond and Ruth Perelman School of Medicine at the University of Pennsylvania, USA.
Different explanations have been offered for the last 40 years to explain how an embryo recognizes its own genome in individual zygote cells, and turns it on. But, it was the Penn team who nailed the mechanism that answers this key question. They found cell size is the key parameter governing genome activation. Cells must achieve a threshold size to initiate large-scale transcription of their own proteins.
By generating and observing miniature embryos, the team saw how changes in cell size controls timing of genome activation.
This work has a number of important implications for understanding how an embryo develops in its earliest days and for the field of developmental biology in general. The Penn team believes their finding could impact how other investigators approach genome activation and screen for maternal factors necessary to control accuracy in early embryo development.
"To gain new insights, zygotic transcription should be measured at a single-cell level. This approach helped us to not overlook the influence of spatial organization of an embryo's cells during maternal-zygotic transition."
Hui Chen, PhD, postdoctoral fellow, Good laboratory.
The 'decision' to initiate a zygote's genome is made at the level of individual cells, not the entire embryo which has changed the Penn team's view of developmental process. He and Chen plan to continue this work by measuring genome activation in zebrafish and mice to see if this new perspective holds true in other species.
Highlights
Quantitative, single-cell imaging of nascent transcription during early embryogenesis
Large-scale ZGA onset is not globally uniform; it follows a stereotypic spatial pattern
Patterned ZGA onset is explained by a cell sizer, not a simple timer or cycle counter
Reducing cell size is sufficient to trigger precocious ZGA in Xenopus embryos
Summary
A defining feature of early embryogenesis is the transition from maternal to zygotic control. This transition requires embryo-wide zygotic genome activation (ZGA), but the extent of spatiotemporal coordination of ZGA between individual cells is unknown. Multiple interrelated parameters, including elapsed time, completed cycles of cell division, and cell size may impact ZGA onset; however, the principal determinant of ZGA during vertebrate embryogenesis is debated. Here, we perform single-cell imaging of large-scale ZGA in whole-mount Xenopus embryos. We find a striking new spatiotemporal pattern of ZGA whose onset tightly correlates with cell size but not with elapsed time or number of cell divisions. Further, reducing cell size induces premature ZGA, dose dependently. We conclude that large-scale ZGA is not spatially uniform and that its onset is determined at the single-cell level, primarily by cell size. Our study suggests that spatial patterns of ZGA onset may be a common feature of embryonic systems.
Authors
Hui Chen PhD, Lily C. Einstein PhD, Shawn C. Little PhD and Matthew C. Good PhD.
Acknowlegements
This work was supported in part by the Burroughs Wellcome Fund, the Charles E. Kaufman Foundation, the March of Dimes, and the National Institution of General Medical Sciences (R35GM12874).
Penn Medicine is one of the world's leading academic medical centers, dedicated to the related missions of medical education, biomedical research, and excellence in patient care. Penn Medicine consists of the Raymond and Ruth Perelman School of Medicine at the University of Pennsylvania (founded in 1765 as the nation's first medical school) and the University of Pennsylvania Health System, which together form a $7.8 billion enterprise.
The Perelman School of Medicine has been ranked among the top medical schools in the United States for more than 20 years, according to U.S. News & World Report's survey of research-oriented medical schools. The School is consistently among the nation's top recipients of funding from the National Institutes of Health, with $425 million awarded in the 2018 fiscal year.
The University of Pennsylvania Health System's patient care facilities include: the Hospital of the University of Pennsylvania and Penn Presbyterian Medical Center--which are recognized as one of the nation's top "Honor Roll" hospitals by U.S. News & World Report--Chester County Hospital; Lancaster General Health; Penn Medicine Princeton Health; and Pennsylvania Hospital, the nation's first hospital, founded in 1751. Additional facilities and enterprises include Good Shepherd Penn Partners, Penn Home Care and Hospice Services, Lancaster Behavioral Health Hospital, and Princeton House Behavioral Health, among others.
Penn Medicine is powered by a talented and dedicated workforce of more than 40,000 people. The organization also has alliances with top community health systems across both Southeastern Pennsylvania and Southern New Jersey, creating more options for patients no matter where they live.
Penn Medicine is committed to improving lives and health through a variety of community-based programs and activities. In fiscal year 2018, Penn Medicine provided more than $525 million to benefit our community.
Return to top of page.
| |
|
Jun 25 2019 Fetal Timeline Maternal Timeline News
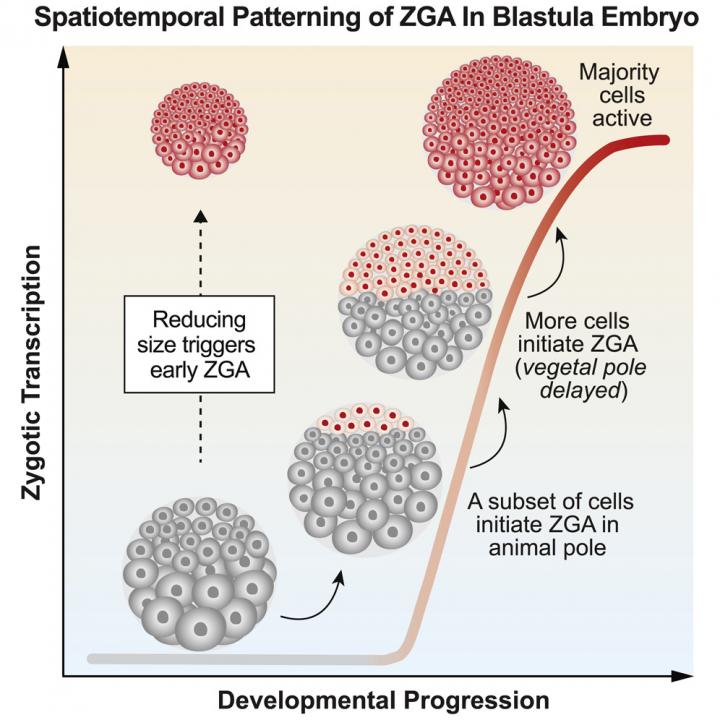 Awakening of the zygote genome over time decreasing individual cell size triggers early embryo transcription. CREDIT Hui Chen, Penn Medicine; Cell Press
|



