|
|
Developmental Biology - Stem Cells
A 3D Model of Early Embryo Development
The earliest stages of life are the most mysterious...
A developing human embryo undergoes a flurry of rapid changes, which are exceedingly difficult to study as they transpire within the confines of a womb.
But with new technology, it might soon be possible to fill important gaps in our understanding of early pregnancy and development. Rockefeller University scientists recently used stem cells to create a 3D model of early embryonic tissues, allowing them to simulate developmental processes as they occur in time and space. They hope this tool, whose utility is demonstrated in Nature Cell Biology, will make it possible to further understand processes guiding embryonic growth, and ultimately lead to innovations promoting healthy pregnancies.
Dimensions of Development
The concept of using stem cells to model early embryonic development was first developed in labs of Ali H. Brivanlou, the Robert and Harriet Heilbrunn Professor, and Eric D. Siggia, the Viola Ward Brinning and Elbert Calhoun Brinning Professor, who published their initial research on the topic in 2014. Despite making several crucial discoveries since that time, Brivanlou and Siggia knew that his system was, in some ways, limited: conventional stem cell models are two dimensional and do not take on the actual shape of an embryo, therefore prohibiting researchers from asking key questions related to its structure.
Scientists are highly interested in the process by which embryos attach to the uterus a crucial first step to a successful pregnancy. And according to Mijo Simunovic, a Simons Junior Fellow in Siggia's lab, it is practically impossible to study this complex phenomenon in a 2-D system.
"Attachment is inherently a 3D problem," says Ali Brivanlou.
To address this issue, Brivanlou, Siggia, and Simunovic used an interdisciplinary approach to develop a 3D model simulating an approximately fourteen-day-old human embryo the stage of development during which a key milestone of embryonic development called "gastrulation" takes place.
"We combined several techniques bioengineering, physics, and developmental biology to create this model," explains Mijo Simunovic PhD, who notes that this research would not have been possible without the unique, longstanding collaboration between the Siggia and Brivanlou labs. "We now have a 3D system that mimics not only the embryo's genetic fingerprint, but also its shape and size."
Of course, it is not enough to make a model that simply looks like a real embryo: it must also act like one. Accordingly, the researchers tested whether their system could simulate one of the most fundamental processes in animal development a phenomenon known as symmetry breaking.
Breaking Symmetry Makes Progress
In its earliest stage, an embryo comprises a symmetrical sphere of cells. Then, after about two weeks, this symmetry begins to disappear as the embryo takes on distinct features that will become various parts of the body.
"Symmetry breaking drives almost everything that happens during embryonic development. Our heads don't look like our feet, and that's because, at some point, the embryo breaks into two parts, anterior and posterior."
Mijo Simunovic PhD, Laboratory of Stem Cell Biology and Molecular Embryology, The Rockefeller University, New York, NY, USA
This breakage is, in fact, the first symmetry breaking of the body axis that takes place during human development, and it transpires just after attachment to the uterus. If the researchers could induce such a breakage in their model, they reasoned, then they would know that their system accurately emulated a real embryo--at least during this key period in developmental time.
To this end, the researchers exposed their model to chemical signals that, in pregnancy, are released by the placenta. Through a series of experiments, they found that the addition of a chemical known as BMP4 reliably prompts symmetry breaking.
"We added BMP4, and two days later one part of the three-dimensional culture became the future posterior, and the opposite part became the future anterior," says Simunovic.
This result has implications beyond elucidating the chemistry of a particular developmental process. Now that scientists can successfully model embryonic events in 3D, extensions of this research may be used in future studies of pregnancy complications, such as unsuccessful attachment.
About 50 to 75 percent of embryos do not attach, creating a huge bottleneck to pregnancy. We don't know why that is, but using this model we may be able to find out."
Mijo Simunovic PhD
This system, Brivanlou notes, could also be used to study inborn diseases:
"We can create 3D embryonic models of genetic conditions, and follow the developmental process in real time. These models can finally advance the understanding of a wide range of diseases for which we currently have no idea where and when things begin to go wrong."
Abstract
Breaking the anteriorposterior symmetry in mammals occurs at gastrulation. Much of the signalling network underlying this process has been elucidated in the mouse; however, there is no direct molecular evidence of events driving axis formation in humans. Here, we use human embryonic stem cells to generate an in vitro three-dimensional model of a human epiblast whose size, cell polarity and gene expression are similar to a day 10 human epiblast. A defined dose of BMP4 spontaneously breaks axial symmetry, and induces markers of the primitive streak and epithelial-to-mesenchymal transition. We show that WNT signalling and its inhibitor DKK1 play key roles in this process downstream of BMP4. Our work demonstrates that a model human epiblast can break axial symmetry despite the absence of asymmetry in the initial signal and of extra-embryonic tissues or maternal cues. Our three-dimensional model is an assay for the molecular events underlying human axial symmetry breaking.
Authors
Mijo Simunovic, Jakob J. Metzger, Fred Etoc, Anna Yoney, Albert Ruzo, Iain Martyn, Gist Croft, Dong Shin You, Ali H. Brivanlou and Eric D. Siggia.
Acknowledgements
M.S. is a Junior Fellow of the Simons Society of Fellows. We were supported by the NIH grant nos R01 HD080699 and R01 GM101653 (to A.H.B. and E.D.S), and NSF PHY grant no. 1502151 (to E.D.S). Imaging was performed at The Rockefeller University Bio-Imaging Resource Center. We thank S. Morgani (MSKCC) and the members of the Siggia and Brivanlou laboratories for their helpful discussions and for critically reading the manuscript.
Return to top of page.
| |
|
Jul 10 2019 Fetal Timeline Maternal Timeline News
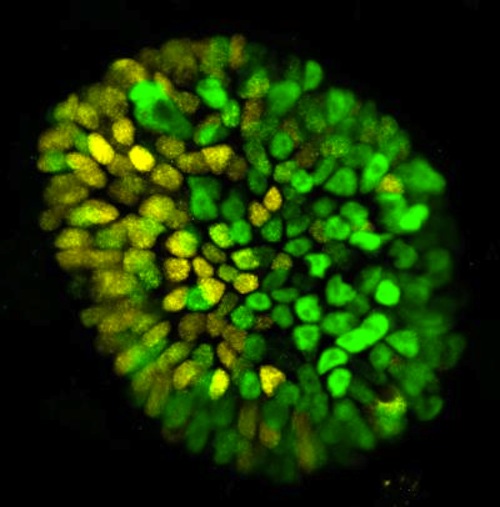 Using a 3D model, researchers were able to simulate the moment in development when the body starts to separate into two distinct halves, here, yellow and green cells. CREDIT Laboratory of Stem Cell Biology and Molecular Embryology at The Rockefeller University.
|



