|
|
Developmental Biology - Heart
What Makes Heart Muscles Contract?
Science finds a protein essential to heart muscle contraction...
A team of scientist led by Dr Enrique Lara Pezzi of the Centro Nacional de Investigaciones Cardio-vasculares (CNIC), has identified SRSF3 RNA-binding protein as essential to heart survival. In a study published in Circulation Research, scientists found reducing the SRSF3 protein critically reduces function of genes needed for heart muscle contraction.
Cardiovascular disease is the leading cause of death around the world. In 2015 alone, cardiovascular disease killed 17.7 million people; 6.7 million of these deaths were by heart attack. Unfortunately, knowledge is limited about the molecular mechanisms regulating progression of myocardial infarction.
Recent developments in technology for sequencing messenger RNA (mRNA) on a massive-scale, now permits identification of gene expression patterns associated with development of distinct tissues such as muscle and heart.
Understanding gene regulation is still limited. In particular, understanding the roles played by RNA-binding proteins (RBP) in the development of heart disease. Dr Lara Pezzi: "In this study, we have investigated the role of the RBP SRSF3 in the heart, which was previously unknown."
So the authors genetically modified mice to allow them to eliminate SRSF3 expression in cardiomyocytes during specific times of development.
First author Dr Paula Ortiz Sánchez found during embryogenesis SRFS3 is highly expressed in cardiomyocyte cells, regulating cell division. "Embryos lacking SRSF3 in cardiomyocytes die," explains Lara Pezzi.
But in the adult heart, "cardiomyocytes barely divide, and SRSF3 expression is much lower, especially after a heart attack indicating the role of SRSF3 in an adult heart must be different. In adults we found reduced function of encoding mRNA proteins in the long, fibrous filaments that slide past each other when a muscle contracts or relaxes (the sarcomeres). This was caused by loss of a chemical modification called the cap found at the 5' end of mRNAs which protects them from being degraded." explains Lara-Pezz PhD.
Further experiments showed that SRSF3 controls processing of mTOR, short for mammalian target of rapamycin. When mTOR is activated, it triggers muscle hypertrophy (increase) through an increase in protein synthesis (how the body turns protein into muscle tissue). When mTOR is on, it helps build muscle and is a major regulator of cell metabolism.
"In the absence of SRSF3, a much shorter version of mTOR is expressed. This shortened form is nonfunctional and causes a series of chemical changes in mTOR-regulated proteins. The outcome is loss of the mRNA cap encoding sarcomere proteins, leading to sarcomere degradation and severe contraction defects seen in SRSF3-deficient mice."
Enrique Lara-Pezz PhD.
The identification of mRNA capping as a mechanism that protects against the development of systolic heart failure could open the way to the development of urgently needed therapeutic tools to combat this disease.
Abstract
Rationale:
RBPs (RNA binding proteins) play critical roles in the cell by regulating mRNA transport, splicing, editing, and stability. The RBP SRSF3 (serine/arginine-rich splicing factor 3) is essential for blastocyst formation and for proper liver development and function. However, its role in the heart has not been explored.
Objective:
To investigate the role of SRSF3 in cardiac function.
Methods and Results:
Cardiac SRSF3 expression was high at mid gestation and decreased during late embryonic development. Mice lacking SRSF3 in the embryonic heart showed impaired cardiomyocyte proliferation and died in utero. In the adult heart, SRSF3 expression was reduced after myocardial infarction, suggesting a possible role in cardiac homeostasis. To determine the role of this RBP in the adult heart, we used an inducible, cardiomyocyte-specific SRSF3 knockout mouse model. After SRSF3 depletion in cardiomyocytes, mice developed severe systolic dysfunction that resulted in death within 8 days. RNA-Seq analysis revealed downregulation of mRNAs encoding sarcomeric and calcium handling proteins. Cardiomyocyte-specific SRSF3 knockout mice also showed evidence of alternative splicing of mTOR (mammalian target of rapamycin) mRNA, generating a shorter protein isoform lacking catalytic activity. This was associated with decreased phosphorylation of 4E-BP1 (eIF4E-binding protein 1), a protein that binds to eIF4E (eukaryotic translation initiation factor 4E) and prevents mRNA decapping. Consequently, we found increased decapping of mRNAs encoding proteins involved in cardiac contraction. Decapping was partially reversed by mTOR activation.
Conclusions:
We show that cardiomyocyte-specific loss of SRSF3 expression results in decapping of critical mRNAs involved in cardiac contraction. The molecular mechanism underlying this effect likely involves the generation of a short mTOR isoform by alternative splicing, resulting in reduced 4E-BP1 phosphorylation. The identification of mRNA decapping as a mechanism of systolic heart failure may open the way to the development of urgently needed therapeutic tools.
Authors
Paula Ortiz-Sánchez, María Villalba-Orero , Marina M. López-Olañeta, Javier Larrasa-Alonso, Fátima Sánchez-Cabo, Carlos Martí-Gómez, Emilio Camafeita, Jesús M. Gómez-Salinero, Laura Ramos-Hernández, Peter J. Nielsen, Jesús Vázquez, Michaela Müller-McNicoll, Pablo García-Pavía, Enrique Lara-Pezz.
Acknowledgements
The authors are grateful to Igor de los Mozos and François McNicoll for assistance with the individual-nucleotide resolution cross-linking and immunoprecipitation experiments and analyses. We thank the CNIC (Centro Nacional de Investigaciones Cardiovasculares) animal facility staff members for mouse work, the Histology Unit for hematoxylin and eosin staining, and the Genomics and Bioinformatics Units for RNA-Seq and analysis.
Sources of Funding
This study was supported by grants from the European Union (CardioNeT-ITN-289600 and CardioNext-ITN-608027 to E. Lara-Pezzi), from the Spanish Ministerio de Economía y Competitividad (RTI2018-096961-B-I00, SAF2015-65722-R, and SAF2012-31451 to E. Lara-Pezzi; BIO2015-67580-P and PGC2018-097019-B-I00 to J. Vázquez), the Spanish Carlos III Institute of Health (CPII14/00027 to E. Lara-Pezzi, RD12/0042/066 to P. García-Pavía and E. Lara-Pezzi, and RD12/0042/0056, PRB2-IPT13/0001-ISCIII-SGEFI/FEDER, ProteoRed to J. Vázquez), the Madrid Regional Government (2010-BMD-2321 Fibroteam to E. Lara-Pezzi). This study was also supported by the Plan Estatal de I+D+I 20132016European Regional Development Fund (ERDF) A way of making Europe, Spain. The CNIC is supported by the Ministerio de Ciencia, Innovación y Universidades (MCNU) and the Pro CNIC Foundation, and is a Severo Ochoa Center of Excellence (SEV-2015-0505).
Disclosures
None.
Footnotes
About the CNIC
The Centro Nacional de Investigaciones Cardiovasculares (CNIC), directed by Dr. Valentín Fuster, is dedicated to cardiovascular research and the translation of knowledge gained into real benefits for patients. The CNIC, recognized by the Spanish government as a Severo Ochoa center of excellence, is financed through a pioneering public-private partnership between the government (through the Carlos III Institute of Health) and the Pro-CNIC Foundation, which brings together 13 of the most important Spanish private companies.
Competing interests
The authors declare no competing interests.
Return to top of page.
| |
|
Jul 19 2019 Fetal Timeline Maternal Timeline News
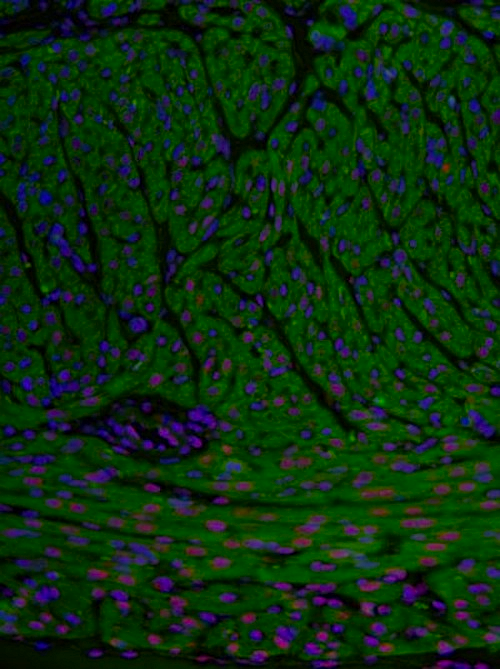 SRSF3 expression in adult cardiomyocytes or heart muscle cells. Pink staining shows SRSF3 functioning in cell nuclei (stained blue). Green stain reflects troponin, a sarcomere protein expressed in cardiomyocytes. CREDIT CNIC
|



