|
|
Developmental Biology - Blood Vessel Formation
Regulating Blood Vessel Formation
How p38 protein regulates formation of new blood vessels...
A study performed at the Institute for Research in Biomedicine (IRB Barcelona) and published in the journal Nature Communications demonstrates how inhibiting production of the p38 protein boosts formation of blood vessels in human [and mouse] colon cancers. Known as angiogenesis, new blood vessel formation is critical in fuelling cancer cells, allowing unlimited growth and eventually spreading metastases.
The team led by Ángel R. Nebreda, head of the Signalling and Cell Cycle Laboratory at IRB Barcelona, shows that p38 activity is important in Mesenchymal Stem Cells (MSCs). MSCs are highly plastic, localize around blood vessels and contribute to tumor development via their response to angiogenesis.
Although it is well known p38 is elevated in cancers, until now little was known about its role in Mesenchymal Stem Cells (MSCs). This study reveals that protein p38 activates new blood vessel formation in tumors through its contribution to MSCs.
"p38 represses angiogenesis [blood vessel formation] specifically through MSCs. Using a mouse model, the study shows inhibition of p38 stimulates formation of new blood vessels both in tumours and during repair of damaged tissue."
Angel R. Nebreda PhD, Institute for Research in Biomedicine (IRB Barcelona), The Barcelona Institute of Science and Technology, Barcelona, Spain.
The study concluded the same mechanisms regulating blood vessel formation, may be of use in optimizing chemotherapy treatments and in diseases involving compromised angiogenesis.
"We hope that the biological knowledge that our work has generated can contribute to the development of more efficient therapies."
Raquel Batlle, postdoctoral fellow, IRB Barcelona and first author of the study.
Abstract
The formation of new blood vessels is essential for normal development, tissue repair and tumor growth. Here we show that inhibition of the kinase p38a enhances angiogenesis in human and mouse colon tumors. Mesenchymal cells can contribute to tumor angiogenesis by regulating proliferation and migration of endothelial cells. We show that p38a negatively regulates an angiogenic program in mesenchymal stem/stromal cells (MSCs), multipotent progenitors found in perivascular locations. This program includes the acquisition of an endothelial phenotype by MSCs mediated by both TGF-ß and JNK, and negatively regulated by p38?. Abrogation of p38a in mesenchymal cells increases tumorigenesis, which correlates with enhanced angiogenesis. Using genetic models, we show that p38? regulates the acquisition of an endothelial-like phenotype by mesenchymal cells in colon tumors and damage tissue. Taken together, our results indicate that p38a in mesenchymal cells restrains a TGF-ß-induced angiogenesis program including their ability to transdifferentiate into endothelial cells.
Authors
Raquel Batlle, Eva Andrés, Lorena Gonzalez, Elisabet Llonch, Ana Igea, Núria Gutierrez-Prat, Antoni Berenguer-Llergo and Angel R. Nebreda.
Acknowledgements
The authors are grateful to Gustavo Leone (Ohio State University, Columbus, USA) for providing FSP1-Cre mice, Ralf Adams (Max Planck Institute for Molecular Biomedicine, Muenster, Germany) for PDGFRB-Cre-ERT2 mice, Eric Brown (University of Pennsylvania School of Medicine, Philadelphia, USA) for UBC-Cre-ERT2 mice, and Manuel Hidalgo (CNIO, Madrid) for PDX samples. We thank the Genomics and Advanced Digital Microscopy Core Facilities of IRB Barcelona for excellent technical assistance, Sebastien Tosi and Anna Lladó for generating the macros used for image quantifications, and Evarist Planet for initial analysis of gene expression data. We are grateful to Eduard Batlle (IRB Barcelona), Ralf Adams (Max Planck Institute Muenster) and Antonio Garcia de Herreros (IMIM Barcelona) for critically reading the manuscript. This work was supported by grants from the European Commission (Advanced ERC 294665), Fundación Olga Torres, Marató-TV3 (20133430), MINECO (SAF2016-81043-R) and AGAUR (2017 SGR-557). IRB Barcelona is the recipient of institutional funding from MINECO (Government of Spain) through the Centres of Excellence Severo Ochoa award and from the CERCA Program of the Catalan Government.
The research has been funded by the European Research Council, the Fundación Olga Torres, the Marató de TV3, the Ministry of Science, Innovation and Universities, and AGAUR (Agency for Management of University and Research Grants).
Return to top of page.
| |
|
Jul 22 2019 Fetal Timeline Maternal Timeline News
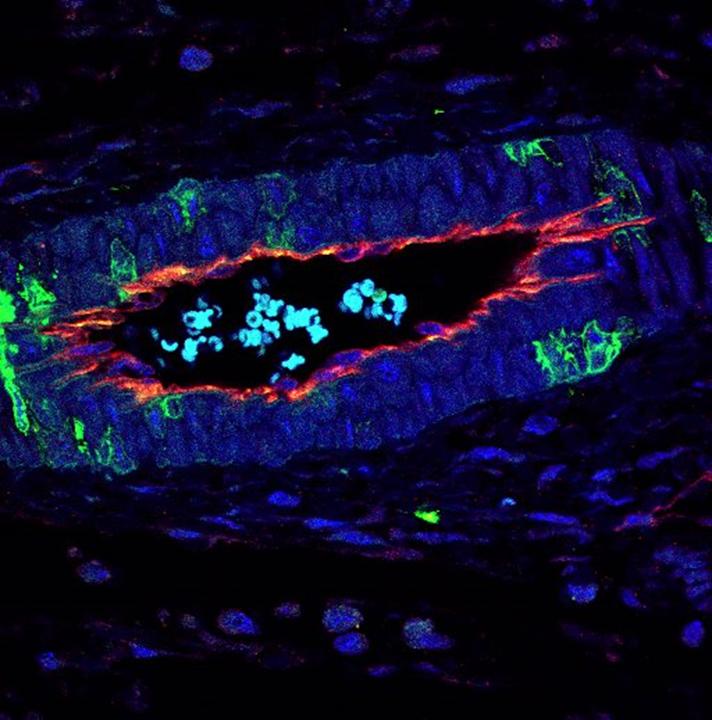 Colon tumors induced in mice were stained with DAPI (BLUE) and immunostained for endothelial marker CD31 (GREEN) and perivascular markers PDGFRB or CD146 (RED). CREDIT Raquel Batlle, IRB Barcelona
|



