|
|
Developmental Biology - Autism
Identified - A Gene Mutation For Autism
A brain protein mutation from a child with autism has been placed into the brains of mice...
For the first time, research is shining a light on a biologic interaction leading to physical behavior observed in autism.
This major finding is published in the Journal of Clinical Investigation and "presents the exciting possibility of a potential mechanistic underpinning - in at least a subset of patients - for some of the altered behaviors observed in ASD and attention deficit hyperactivity disorder, or ADHD," explains Aurelio Galli PhD, Professor of Surgery, University of Alabama at Birmingham.
The research was led by both corresponding authors Aurelio Galli and Mark Wallace PhD, a neurobiologist and dean at Vanderbilt University.
The brain protein studied is DAT, a dopamine transporter.
Certain brain neurons release dopamine, a neurotransmitter, from ends of each axon extending from the neuron cell.
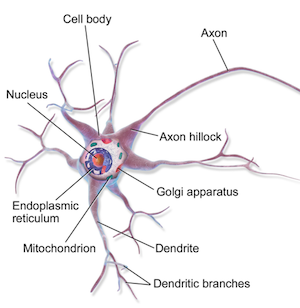 The function of the axon is to transmit information to different neurons, muscles, and glands. The function of the axon is to transmit information to different neurons, muscles, and glands.
Normally, dopamine crosses the synapse junction between an axon and a neighboring neuron, triggering a response in that neuron. However, if DAT sits within the membrane of a neuron, that neuron reuptakes dopamine pumping it back across the synapse junction, terminating any neural response.
In the striatum region of the brain - dopamine is critical in regulating motor activity, motivation, attention and reward.
Based on dopamine's critical role in nerve signal transmission, abruptly ending transmissions presents as neuropsychiatric disorder such as: Parkinson's disease, drug abuse of heroin - cocaine - speed - nicotine and more; bipolar disorder; Attention Deficit Hyperactivity Disorder (ADHD); and recently Autism Spectrum Disorder (ASD).
Galli, Wallace and colleagues found a mutation in the gene for human DAT in a child with ASD. This mutation generates a substitution at amino acid 356 of DAT, a change from threonine to methionine, calling it DAT T356M.
A previous study led by Galli and Eric Gouaux PhD, professor, Oregon Health & Science University, introduced the mutation into a fruit fly DAT. Now, flies carrying DAT T356M acted abnormally with increased locomotor activity, fear, repetitive activity and altered social interaction, much like autism impairments in humans. Bacterial studies suggested that DAT T356M is flipped inside-out compared to normal DAT, so that DAT T356M anomalously pumps dopamine out of a cell rather than into a cell.
Now Galli, Wallace, and MD PhD student Gabriella DiCarlo report the first study on DAT T356M as found in a mammalian brain.
Mice that have two copies of the DAT T356M gene (homozygous) mutation showed severe behavior changes resembling human ASD and ADHD with significant alterations in their brain physiology. ADHD exists simultaneously with ASD - a comorbidity. In contrast, no changes were seen in mice that had only one copy of the DAT T356M gene mutation, when compared to normal mice.
Mice with DAT T356M showed a loss of socialbility, a loss of social dominance and a diminished interest in burying marbles which is an innate behavior of lab mice, motivated by their desire to bury food. Mice with DAT T356M had repetitive rearing behaviors and enhanced ability to keep balance on a rotating rod linked to their repetitive behavior. However, when treated with two different compounds that block DAT activity, their hyperactive behavior decreased.
"Future work should aim to determine whether blocking DAT may eliminate or alleviate the more complex behavioral changes observed in DAT T356M animals."
Aurelio Galli PhD, D.Sc, Professor of Surgery and Director for Gastrointestinal Biology Research. The University of Alabama at Birmingham, Alabama, USA.
Abstract
The precise regulation of synaptic dopamine (DA) content by the DA transporter (DAT) ensures the phasic nature of the DA signal, which underlies the ability of DA to encode reward prediction error, thereby driving motivation, attention, and behavioral learning. Disruptions to the DA system are implicated in a number of neuropsychiatric disorders, including attention deficit hyperactivity disorder (ADHD) and, more recently, autism spectrum disorder (ASD). An ASD-associated de novo mutation in the SLC6A3 gene resulting in a threonine-to-methionine substitution at site 356 (DAT T356M) was recently identified and has been shown to drive persistent reverse transport of DA (i.e., anomalous DA efflux) in transfected cells and to drive hyperlocomotion in Drosophila melanogaster. A corresponding mutation in the leucine transporter, a DAT-homologous transporter, promotes an outward-facing transporter conformation upon substrate binding, a conformation possibly underlying anomalous DA efflux. Here, we investigated in vivo the impact of this ASD-associated mutation on DA signaling and ASD-associated behaviors. We found that mice homozygous for this mutation displayed impaired striatal DA neurotransmission and altered DA-dependent behaviors that correspond with some of the behavioral phenotypes observed in ASD.
Authors
Gabriella E. DiCarlo, Jenny I. Aguilar, Heinrich J. G. Matthies, Fiona E. Harrison, Kyle E. Bundschuh, Alyssa West, Parastoo Hashemi, Freja Herborg, Mattias Rickhag, Hao Chen, Ulrik Gether, Mark T. Wallace and Aurelio Galli.
Acknowledgements
The authors acknowledge financial support for this study from the National Science Foundation-National Institutes of Environmental Health Sciences Oceans and Human Health Program (OCE-1321612).
Support came from National Institutes of Health grants DA038058, DA35263, MH115535, MH114316, MH106563 and GM007347.
Return to top of page.
| |
|
Jul 29, 2019 Fetal Timeline Maternal Timeline News
 Mice with DAT T356M had repetitive rearing behaviors and enhanced ability to keep balance on a rotating rod linked to their repetitive behavior. However, when treated with two different compounds that block DAT activity, their hyperactive behavior decreased.
|




