|
|
Developmental Biology - Opto-ribogenetics
Controlling RNA With Light!
Researchers use blue light to regulate the activity of RNA molecules...
Messenger RNA molecules contain genetic information controlling how molecules organize into proteins and provide for the growth of tissues. Now, biochemists at the University of Bayreuth and the University of Bonn, Germany, have discovered a way to intervene and possibly regulate this process using an actinobacteria that contains a protein which binds to RNA molecules under blue light and can deactivate the whole process.
In principle, it is possible to switch RNA-controlled protein synthesis on and off using light, not just in bacteria but even in mammalian and human cells.
The findings are published in Nature Chemical Biology are the basis for a new field of research: opto-ribogenetics.
For some time now, light signals have been used to alter transcription of genetic information and the proteins created and directed by RNA (Ribonucleic Acids) molecules at the DNA level. This optogenetic approach is now part of a well-established method of molecular and cell biology. However, a new study now shows for the first time how the interaction between RNA and specific proteins can be influenced by light.
Gene expression in bacteria now and going forward can be controlled directly at the level of RNA molecules. The researchers led by Prof. Dr. Andreas Möglich in Bayreuth and Prof. Dr. Günter Mayer in Bonn have demonstrated that this mechanism can be transferred to mammalian cells.
"Over the next few years, we will extend the light-controlled regulation to various cellular processes involving RNA. The resulting tools, which have not been available to date, will greatly advance the investigation of central cellular processes. The foundation stone for optoribogenetics, a new complement to optogenetics, has now been laid."
Andreas Möglich PhD, Professor, Chairman of Biochemistry, Bayreuth Center for Biochemistry and Molecular Biology; North-Bavarian Nuclear Magnetic Resonance Center; Universty of Bayreuth, Bayreuth, Germany.
Search for a candidate protein reacting to light
The starting point of the research work was the hunt for a bacterial photoreceptor protein able to change its own binding behaviour in relation to RNA under the influence of light. The scientists searched the existing sequence databases and found what they were looking for. Bacteria of the species Nakamurella multipartita contain a protein with a conspicuous tripartite architecture: three different sections or "domains" called (1) PAS, (2) ANTAR and (3) LOV, are arranged one after the other in an unusual sequence.
Shown in cooperation with the research group of Prof. Dr. Robert Bittl at Freie Universität Berlin, the LOV photosensor domain reacts to blue light and transmits the signals to the ANTAR domain. The ANTAR domain then changes its structure so that RNA molecules are bound and inaccessible, no longer available for gene function. Their genetic information cannot be used to make proteins.
Only when the blue light irradiation ceases, and the ANTAR domain returns to its normal structure, does interaction with RNA stop. Now the RNA becomes active again. The researchers first established and demonstrated this process using RNA aptamers. These are small RNA molecules with a hairpin-like structure that can enter the structure of the ANTAR domain, which is opened under blue light, and are bound there. Mayer: "Aptamers work in modular fashion: They can be linked to other units like a building block system."
The scientists also tested their new research approach on eukaryotic cells into which they had previously introduced the bacterial protein and the RNA aptamers. In these cells, too, the structural changes triggered by blue light lead to messenger RNA molecules binding to the protein and, in this state, suspending gene expression.
"We now have a light switch with which the cellular activity of different RNA molecules can be specifically switched on and off," explains Prof. Dr. Günter Mayer from the LIMES Institute at the University of Bonn.
His colleague from Bayreuth, Prof. Dr. Andreas Möglich, adds: "The approach to light-regulated control can in principle be transferred to numerous other RNA-based processes, such as the processing of micro-RNAs and the associated phenomenon of gene silencing."
In future studies, the two research groups hope to investigate the extent to which this new mechanism can be used in model organisms to control gene expression and other processes.
Abstract
Sensory photoreceptor proteins underpin light-dependent adaptations in nature and enable the optogenetic control of organismal behavior and physiology. We identified the bacterial light-oxygen-voltage (LOV) photoreceptor PAL that sequence-specifically binds short RNA stem loops with around 20 nM affinity in blue light and weaker than 1 µM in darkness. A crystal structure rationalizes the unusual receptor architecture of PAL with C-terminal LOV photosensor and N-terminal effector units. The light-activated PALRNA interaction can be harnessed to regulate gene expression at the RNA level as a function of light in both bacteria and mammalian cells. The present results elucidate a new signal-transduction paradigm in LOV receptors and conjoin RNA biology with optogenetic regulation, thereby paving the way toward hitherto inaccessible optoribogenetic modalities.
Authors
Anna M. Weber, Jennifer Kaiser, Thea Ziegler, Sebastian Pilsl, Christian Renzl, Lisa Sixt, Georg Pietruschka, Sébastien Moniot, Ankana Kakoti, Marc Juraschitz, Stefanie Schrottke, Laura Lledo Bryant, Clemens Steegborn, Robert Bittl, Günter Mayer and Andreas Möglich.
Acknowledgements
The authors thank members of the Mayer and Möglich laboratories for discussion; H. Ruwe and C. Schmitz-Linneweber (HU Berlin) for help in the initial analysis of nucleic-acid binding; M. Humenik for assistance with SECMALS. Funding through a Sofja-Kovalevskaya Award of the Alexander-von-Humboldt Foundation (to A.M.); a Consolidator grant no. 615381 by the European Research Council (to G.M.) and Deutsche Forschungsgemeinschaft (nos. MO2192/6-1, MA3442/5-1, SFB-1089/P1 and Sfb-1078/B4) is gratefully acknowledged.
Return to top of page.
| |
|
Sep 2 2019 Fetal Timeline Maternal Timeline News
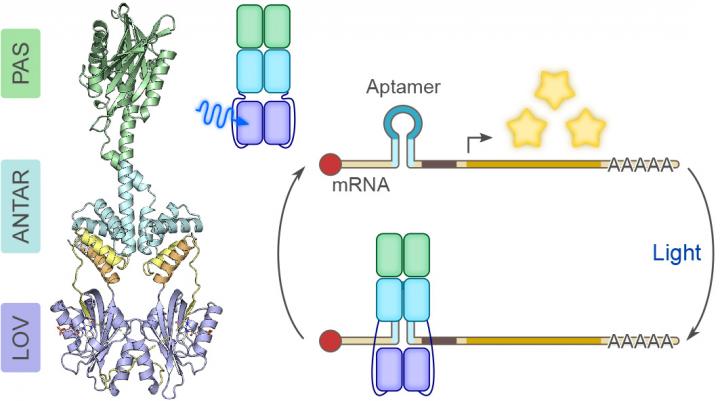 The photoreceptor PAL binds to short ribonucleic acids, made visible by blue light irradiation. This light-controlled activity can be used to regulate all RNA-based processes.
CREDIT Andreas Möglich/University of Bayreuth, Bonn, Germany.
|



