|
|
Developmental Biology - Muscle Renewal
Target For Healing Old Muscles?
Removing protein that suppresses renewal of muscle stem cells - allows muscles to regenerate...
Discovered: Possible therapeutic target for slow healing of aged muscles
New work could reveal a potential pathway for therapeutic targeting to combat muscle degeneration in the elderly
Washington, DC An age-related decline in recovery from muscle injury can be traced to a protein that suppresses the special ability of muscle stem cells to build new muscles, according to work from a team of current and former Carnegie biologists led by Chen-Ming Fan of the Department of Biology, Johns Hopkins University, Baltimore, and the Department of Embryology, Carnegie Institution for Science, also in Baltimore, Maryland, USA. The work is published in Nature Metabolism.
Skeletal muscles have a tremendous capacity to make new muscle tissue from special muscle stem cells. These "blank" cells are not only good at making muscles but also at generating more of themselves, a process called self-renewal.
But this amazing ability diminishes with age. Over time resulting in poorer regeneration.
The research team, including Carnegie's Liangji Li, Michelle Rozo, Sibiao Yue, Xiaobin Zheng, and Frederick Tan, as well as Christoph Lepper formerly of Carnegie now at the Ohio State University, found a protein called GAS1 is the culprit for this age-related decline.
"Encoded by the growth-arrest specific gene, the GAS1 protein lives up to its name, accelerating the functional decline of muscle stem cells."
Liangji Li, Department of Biology, Johns Hopkins University, Baltimore, Maryland and Department of Embryology, Carnegie Institution for Science, Baltimore, Maryland, USA and lead author.
They discovered the protein is found in only a small number of young muscle stem cells, but is present in all aged muscle stem cells. Tinkering with muscle stem cells to express GAS1 in the entire young stem cell population resulted in diminished regeneration. By contrast, removing GAS1 from aged muscle stem cells rejuvenated them to a youthful state that supported robust regeneration.
They also discovered that GAS1 inhibits another protein, a cell-surface receptor called RET, which they showed to be necessary for muscle stem cell renewal. The more GAS1 protein is present, the more RET's function is reduced.
The inhibition of RET by GAS1 could be reversed by the third protein called GDNF, which binds to and activates RET. Indeed, when GDNF was injected directly into muscles of aged mice, muscle stem cell function and regeneration were restored.
Fan continues: "With a rapidly aging population, issues like muscle deterioration are an increasing societal challenge. Our work could reveal a potential pathway for therapeutic targeting to combat muscle degeneration in the elderly."
Abstract
Muscle undergoes progressive weakening and regenerative dysfunction with age due in part to the functional decline of skeletal muscle stem cells (MuSCs). MuSCs are heterogeneous, but whether their gene expression changes with age and the implication of such changes are unclear. Here we show that in mice, growth arrest-specific gene 1 (Gas1) is expressed in a small subset of young MuSCs, with its expression progressively increasing in larger fractions of MuSCs later in life. Overexpression of Gas1 in young MuSCs and inactivation of Gas1 in aged MuSCs support that Gas1 reduces the quiescence and self-renewal capacity of MuSCs. GAS1 reduces RET signalling, which is required for MuSC quiescence and self-renewal. Indeed, we show that the RET ligand, glial-cell-line-derived neurotrophic factor can counteract GAS1 by stimulating RET signalling and enhancing MuSC self-renewal and regeneration, thus improving muscle function. We propose that strategies aimed at targeting this pathway can be exploited to improve the regenerative decline of MuSCs.
Authors
Liangji Li, Michelle Rozo, Sibiao Yue, Xiaobin Zheng, Frederick J. Tan, Christoph Lepper and Chen-Ming Fan.
Acknowledgments
The authors thank Fan laboratory members and Y. Zheng for comments; E. Dikovsky for the mouse facility team; and S. Satchell for technical assistance. They especially thank T. Cheung for sharing the FACS protocol. L.L. is supported by the Carnegie Institution of Washington. M.R. was supported by a predoctoral fellowship from the NIH (no. HD075345). C.-M.F. is supported by the NIH (grant nos. R01AR060042, R01AR071976 and R01AR072644) and the Carnegie Institution of Washington.
Return to top of page.
| |
|
Oct 8 2019 Fetal Timeline Maternal Timeline News
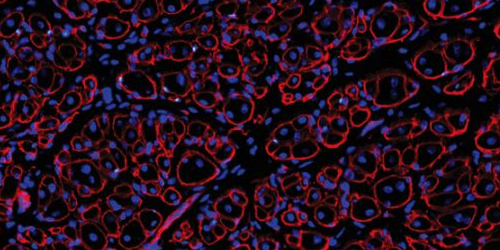 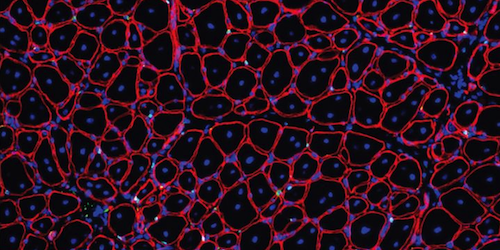 Treatment with GDNF repairs aged muscle stem cells & supports robust muscle regeneration: TOP - Small muscle fibers (red shapes with blue nuclei), made by old muscle stem cells. BOTTOM - Increased muscle fiber size/ muscle stem cell numbers (GREEN DOTS) after GDNF. CREDIT Image provided courtesy of Liangji Li, Carnegie Institution of Washington, DC.
|




