|
|
Developmental Biology - Cell Change
MYH9 Gene Mutations Cause Numerous Disorders
Using live imaging, researchers watch cells move and generate numerous changes inside living tissue...
There is new information on how motor proteins generate change inside living tissue.
Myosins are motor proteins that convert chemical energy into mechanical force. Myosin II specifically generates force essential to drive cell movement and shape change giving rise to tissue structures. Altough researchers know gene mutations in non-muscle myosin II can lead to severe congenital defects in adults, and diseases such as blood platelet dysfunction, nephritis, and deafness they don't fully understand how myosin activity is altered into specific physiologic change.
A team of researchers led by Karen Kasza, Clare Boothe Luce Assistant Professor of Mechanical Engineering, uses drosophila [fruit fly] embryos to model human disease mutations affecting myosin motor activity. Through in vivo [live] imaging and biophysical analysis, Kasza's team demonstrated they could engineer human MYH9-related disease mutations into drosophila myosin II and alter drosophila to fail in their ability to drive rapid cell movements.Failures which resulted in defects in epithelial morphogenesis. Her study is the first to demonstrate how these mutations result in slower cell movements in vivo. It was published October 15, 2019, in PNAS.
Karen E. Kasza PhD, Howard Hughes Medical Institute and Developmental Biology Program, Sloan Kettering Institute, New York, NY; Department of Mechanical Engineering, Columbia University, New York, NY, USA and lead author.
Because there are so many similarities between the myosin II protein in humans and in fruit flies, Kasza's approach was to start by tackling how to "watch" the effects of myosin II mutations in fruit flies. Her group engineered the human disease mutations into fruit fly myosin and then observed how this affected the behaviors of the proteins, cells, and tissues in the organism.
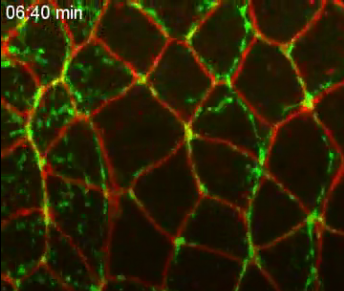
The team used high-resolution confocal fluorescence imaging of the process, together with biophysical laser nano-dissection, to measure forces generated by mutated myosin II motor proteins in vivo.
Kasza found the mutated myosin II motor proteins actually migrated to their proper places inside cells and was able to generate force. However, the fine-scale organization of the myosin proteins and speed of their movement inside cells was slower than in normal wild-type myosin protein. This slower cellular movement brought about shape abnormalities in drosophila embryos during development.
"By 'watching' how cells move and generate forces inside living tissues, we've uncovered new clues as to why mutations in the MYH9 gene cause a broad spectrum of disorders in humans. This mechanistic understanding will help us better understand these diseases and could lead to new diagnostic or therapeutic strategies down the road." explains Kasza.
The researchers are now working on new approaches to very precisely manipulate the forces generated by myosin motors inside living cells and tissues. These new tools will help the team to uncover how mechanical forces influence biochemical processes that control cell movements and cell fate. These studies will be essential to better understanding how dysregulation of mechanical forces contributes to disease.
Significance
Cells require forces generated by the contractile actomyosin cytoskeleton to remodel and shape tissues. In humans, mutations in the myosin II motor are associated with a wide range of diseases, but the cellular mechanisms that underlie disease physiologies are not well understood. Using in vivo imaging and biophysical approaches, we show that engineering human disease mutations in fruit fly myosin II produces proteins with altered organization and dynamics that fail to drive rapid cell movements, resulting in abnormalities in embryo shape. Because many genes and cell behaviors are shared between fruit flies and humans, these results may reveal mechanisms underlying myosin II-related diseases in humans.
Abstract
The nonmuscle myosin II motor protein produces forces that are essential to driving the cell movements and cell shape changes that generate tissue structure. Mutations in myosin II that are associated with human diseases are predicted to disrupt critical aspects of myosin function, but the mechanisms that translate altered myosin activity into specific changes in tissue organization and physiology are not well understood. Here we use the Drosophila embryo to model human disease mutations that affect myosin motor activity. Using in vivo imaging and biophysical analysis, we show that engineering human MYH9-related disease mutations into Drosophila myosin II produces motors with altered organization and dynamics that fail to drive rapid cell movements, resulting in defects in epithelial morphogenesis. In embryos that express the Drosophila myosin motor variants R707C or N98K and have reduced levels of wild-type myosin, myosin motors are correctly planar polarized and generate anisotropic contractile tension in the tissue. However, expression of these motor variants is associated with a cellular-scale reduction in the speed of cell intercalation, resulting in a failure to promote full elongation of the body axis. In addition, these myosin motor variants display slowed turnover and aberrant aggregation at the cell cortex, indicating that mutations in the motor domain influence mesoscale properties of myosin organization and dynamics. These results demonstrate that disease-associated mutations in the myosin II motor domain disrupt specific aspects of myosin localization and activity during cell intercalation, linking molecular changes in myosin activity to defects in tissue morphogenesis.
Authors
Karen E. Kasza, Sara Supriyatno and Jennifer A. Zallen.
The authors declare no financial or other conflicts of interest.
Acknowledgments
Sloan Kettering Institute
The study was supported by NIH/NIGMS R01 grant GM102803 to JAZ. KEK holds a Career Award at the Scientific Interface from the Burroughs Wellcome Fund, a Clare Boothe Luce Professorship, and a Packard Fellowship. JAZ is an investigator of the Howard Hughes Medical Institute.
Columbia Engineering, based in New York City, is one of the top engineering schools in the U.S. and one of the oldest in the nation. Also known as The Fu Foundation School of Engineering and Applied Science, the School expands knowledge and advances technology through the pioneering research of its more than 220 faculty, while educating undergraduate and graduate students in a collaborative environment to become leaders informed by a firm foundation in engineering. The School's faculty are at the center of the University's cross-disciplinary research, contributing to the Data Science Institute, Earth Institute, Zuckerman Mind Brain Behavior Institute, Precision Medicine Initiative, and the Columbia Nano Initiative. Guided by its strategic vision, "Columbia Engineering for Humanity," the School aims to translate ideas into innovations that foster a sustainable, healthy, secure, connected, and creative humanity..
Return to top of page.
| |
|
Nov 4 2019 Fetal Timeline Maternal Timeline News
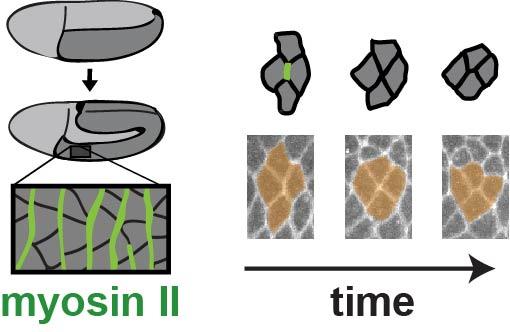
Left: A schematic of myosin-driven body axis elongation in fruit fly embryo. Arrow points to cell shape changes from disease-associated mutations in (GREEN) 'myosin II motor protein.'
Right: Alterations in myosin motor proteins slow cell movement in living tissue. CREDIT Karen Kasza/Columbia Engineering & Sara Supriyatno/Sloan Kettering Institute.
|