|
|
Developmental Biology - Fertility
Tiny Hydractinia Produces Unlimited Eggs & Sperm
Research on Hydractinia might provide clues to human reproduction...
A little-known ocean-dwelling creature most commonly found growing on dead hermit crab shells may sound like an unlikely subject for study, except for its rare ability to make eggs and sperm throughout its entire life.
The Hydractinia produces germ cells (cells that are precursors to eggs and sperm) nonstop throughout its life. Studying this animal's unique ability could give us insight into how our human reproductive system developed, and possibly how human reproductive errors and diseases occur.
"By sequencing and studying the genomes of simpler organisms [such as Hydractinia] which are easy to manipulate in the lab, we've been able to tease out insights regarding germ cell fate - knowledge that may help us understand processes underlying reproductive disorders in humans."
Andy Baxevanis PhD, Director, National Human Genome Research Institute's (NHGRI is a part of the National Institutes of Health) Computational Genomics Unit and co-author of the paper.
The study is published in the journal Science, by collaborators at NHGRI, the National University of Ireland, Galway, and the Whitney Laboratory for Marine Bioscience at the University of Florida.
Researchers described how activating the Tfap2 gene in Hydractinia adult stem cells turned those cells into germ cells [egg or sperm] in an endless repeating cycle.
By comparison, most mammals generate a specific number of germ cells only once in their lifetime. Eggs and sperm may form from those germ cells for a period of time, but always in limited amounts and not for an entire human lifetime.
Hydractinia live in colonies and are closely related to jellyfish and corals. Although dissimilar to humans physically, a Hydractinia's genome [its entire gene makeup] contains a surprisingly large number of genes similar to human disease genes, making it a useful animal model for study of human health issues.
Hydractinia have specialized sexual polyps that produce eggs and sperm, making those polyps functionally similar to our human gonads.
During human embryo development, a small pool of germ cells eventually become eggs or sperm. These are set aside and will produce all sperm or eggs from that individual. Loss of these germ cells for any reason results in that person's sterility.
Recently, a separate study by Andreas Baxevanis and Christine Schnitzler sequenced the entire Hydractinia genome for the first time looking for clues as to what marked differences exist between humans and this distant relative.
"Having this kind of high-quality, whole-genome sequence data in hand allowed us to quickly narrow down the search for a specific gene or genes that tell Hydractinia's stem cells to become germ cells," explained Baxevanis. They then compared the behavior of genes in the feeding and sexual structures of Hydractinia to find the Tfap2 gene is much more active in Hydractinia sexual polyps than in their feeding polyps in both males and females.
The study confirmed Tfap2 is indeed the switch controlling the process of perpetual germ cell production.
Using the CRISPR-Cas9 gene-editing technique to remove Tfap2 from Hydractinia, confirmed Tfap2 stopped germ cell formation. Accordingly, the research also found Tfap2 gene regulates germ cell production in humans.
In humans, germ cells (egg and sperm) are separated from non-germ cells early in development, and germ cell production is for less than a lifetime. Yet despite the enormous evolutionary distance between Hydractinia and humans, both share a key gene that changes stem cells into germ cells.
Abstract
Clonal animals do not sequester a germ line during embryogenesis. Instead, they have adult stem cells that contribute to somatic tissues or gametes. How germ fate is induced in these animals, and whether this process is related to bilaterian embryonic germline induction, is unknown. We show that transcription factor AP2 (Tfap2), a regulator of mammalian germ lines, acts to commit adult stem cells, known as i-cells, to the germ cell fate in the clonal cnidarian Hydractinia symbiolongicarpus. Tfap2 mutants lacked germ cells and gonads. Transplanted wild-type cells rescued gonad development but not germ cell induction in Tfap2 mutants. Forced expression of Tfap2 in i-cells converted them to germ cells. Therefore, Tfap2 is a regulator of germ cell commitment across germ linesequestering and germ linenonsequestering animals.
Conserved Gene Specifies Germ Cell
Germ cells are the exclusive progenitors of gametes. In most studied animals, including humans, germ cells are produced only once during embryogenesis and are not replenished in adult life. DuBuc et al. studied germ cell induction in the clonal cnidarian Hydractinia symbiolongicarpus, an animal that forms germ cells continuously in adult life from stem cells that also generate somatic cells. A single transcription factor is capable of converting the animal's adult stem cells to germ cells. A similar gene also controls germ cell induction in mammalian embryos, but its action there is limited to a single event in early embryogenesis.
Authors
Timothy Q. DuBuc, Christine E. Schnitzler, Eleni Chrysostomou, Emma T. McMahon, Febrimarsa, James M. Gahan, Tara Buggie, Sebastian G. Gornik, Shirley Hanley, Sofia N. Barreira, Paul Gonzalez, Andreas D. Baxevanis and Uri Frank.
Return to top of page.
|
|
Feb 18 2020 Fetal Timeline Maternal Timeline News
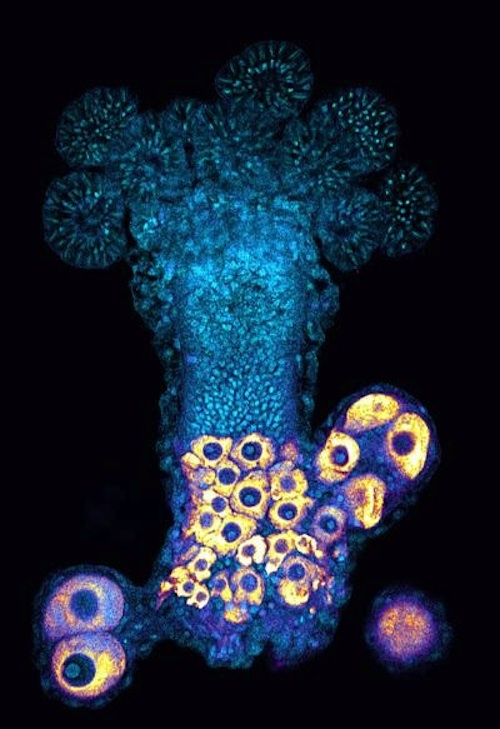 Female Hydractinia emitting from a specialized sexual structure which functions similarly to the human ovary yellow eggs (oocytes) each with a turquoise nucleus. Male Hydractinia have a structure functioning similarly to the human testis. CREDIT Timothy DuBuc PhD, Swarthmore College
|
|



