|
|
Developmental Biology - Infant Immune System
CCL2 Molecule Important To Autoimmune Defense
Medications used in adults and children with different forms of autoimmune inflammatory arthritis or lupus are now being examined in response to coronavirus infections...
A study by investigators at Hospital for Special Surgery (HSS) has discovered a molecule in the lymphatic system with the potential to play a role in autoimmune disease. The study was published online March 20, 2020 in the journal Science Immunology.
Lead investigator Theresa Lu, MD, PhD, in the Autoimmunity and Inflammation Program at the HSS Research Institute, and colleagues launched the study to gain a better understanding of how the immune system works.
A healthy immune system defends the body against disease and infection. When someone has an autoimmune disease, the immune system malfunctions and the body mistakenly attacks healthy cells, tissues and organs.
Rheumatoid arthritis, lupus and scleroderma are examples of autoimmune diseases. If scientists can elucidate the underlying mechanism causing autoimmune and inflammatory conditions, they may be able to correct immune system flaws leading to disease.
The study focused on lymphoid tissues, which house immune cells and are sites of immune cell activation.
Lymphoid tissues include tonsils, spleen, lymph nodes and structures such as fibroblasts and blood vessels. The latter were thought mainly to provide infrastructure for immune cells, but recent advances show they actively affect cell response. Multiple populations of fibroblasts have different functions, according to Dr. Lu.
"One fibroblast population expressed a molecule called CCL2 in the area of antibody secreting immune cells, called plasma cells. We focused on CCL2-expressing fibroblasts to see if they regulate plasma cell function.
It was found to limit the magnitude of plasma cell responses - acting on an intermediary cell to reduce plasma cell survival. This was surprising, as CCL2 can also promote inflammation.
This underscores the multiple functions that any molecule can have in different contexts."
Theresa Lu MD PhD, Senior Scientist , Autoimmunity and Inflammation Program, HSS Research Institute, and lead investigator.
These findings have implications for better understanding autoimmune diseases, according to Dr Lu. Plasma cells in autoimmune diseases generate autoantibodies that then deposit and cause inflammation in organs such as the kidneys and skin.
"By understanding that plasma cells can be controlled by this subset of fibroblasts, we can study them to see if they are not working properly in autoimmune and inflammatory diseases and search for ways to correct malfunctions, so they are less likely to cause disease.
As the immune system is central to how well our bodies function, what we learn about manipulating fibroblast cells can help the biomedical community better understand how to treat related processes such as healing musculoskeletal injury, fighting cancer and fighting infections.
For example those medications used in adults and children with different forms of autoimmune inflammatory arthritis or lupus are now being examined in the setting of coronavirus infections.
We all learn from each other."
Theresa Lu MD PhD
Dr. Lu's lab and her colleagues have been studying the vasculature and fibroblasts of lymph nodes for 16 years. Dragos Dasoveanu PhD, is first author of the current work, conducting research at HSS along with collaborators in New York, Switzerland and Australia.
Sending messages to plasma cells
Lymph node stromal cells serve as a platform that facilitates functional interactions between distinct immune cell types. Here, Dasoveanu et al. have examined the role of fibroblastic reticular cells (FRCs), a type of lymph node stromal cells, in regulating the survival of antibody-producing plasma cells. They report FRCs to be a critical source of chemokine ligand 2 (CCL2), and that CCL2 produced by FRCs tempers the expansion of plasma cells. Plasma cells do not express the CCL2 receptor CCR2; rather, CCR2-expressing monocytes respond to CCL2 by generating molecules that relay signals from the FRCs to the plasma cells. The study adds to the growing appreciation of the roles of lymph node stromal cells in fine-tuning adaptive immune responses.
Abstract
Nonhematopoietic stromal cells in lymph nodes such as fibroblastic reticular cells (FRCs) can support the survival of plasmablasts and plasma cells [together, antibody-forming cells (AFCs)]. However, a regulatory function for the stromal compartment in AFC accumulation has not been appreciated. Here, we show that chemokine ligand 2 (CCL2)expressing stromal cells limit AFC survival. FRCs express high levels of CCL2 in vessel-rich areas of the T cell zone and the medulla, where AFCs are located. FRC CCL2 is up-regulated during AFC accumulation, and we use lymph node transplantation to show that CCL2 deficiency in BP3+ FRCs and lymphatic endothelial cells increases AFC survival without affecting B or germinal center cell numbers. Monocytes are key expressers of the CCL2 receptor CCR2, as monocyte depletion and transfer late in AFC responses increases and decreases AFC accumulation, respectively. Monocytes express reactive oxygen species (ROS) in an NADPH oxidase 2 (NOX2)dependent manner, and NOX2-deficient monocytes fail to reduce AFC numbers. Stromal CCL2 modulates both monocyte accumulation and ROS production, and is regulated, in part, by manipulations that modulate vascular permeability. Together, our results reveal that the lymph node stromal compartment, by influencing monocyte accumulation and functional phenotype, has a regulatory role in AFC survival. Our results further suggest a role for inflammation-induced vascular activity in tuning the lymph node microenvironment. The understanding of stromal-mediated AFC regulation in vessel-rich environments could potentially be harnessed to control antibody-mediated autoimmunity.
Authors
Dragos C. Dasoveanu, Hyeung Ju Park, Catherine L. Ly, William D. Shipman, Susan Chyou, Varsha Kumar, David Tarlinton, Burkhard Ludewig, Babak J. Mehrara and Theresa T. Lu.
Acknowledgements
Hospital for Special Surgery (HSS) is the world's leading academic medical center focused on musculoskeletal health. It is nationally ranked No. 1 in orthopedics (for the tenth consecutive year), No. 3 in rheumatology by U.S. News & World Report (2019-2020), and named a leader in pediatric orthopedics by U.S. News & World Report "Best Children's Hospitals" list (2019-2020). Founded in 1863, the Hospital has one of the lowest infection rates in the country and was the first in New York State to receive Magnet Recognition for Excellence in Nursing Service from the American Nurses Credentialing Center four consecutive times. The global standard total knee replacement was developed at HSS in 1969. An affiliate of Weill Cornell Medical College, HSS has a main campus in New York City and facilities in New Jersey, Connecticut and in the Long Island and Westchester County regions of New York State. In addition, HSS will be opening a new facility in Florida in early 2020. In 2018, HSS provided care to 139,000 patients and performed more than 32,000 surgical procedures, and people from all 50 U.S. states and 80 countries travelled to receive care at HSS. There were more than 37,000 pediatric visits to the HSS Lerner Children's Pavilion for treatment by a team of interdisciplinary experts. In addition to patient care, HSS leads the field in research, innovation and education. The HSS Research Institute comprises 20 laboratories and 300 staff members focused on leading the advancement of musculoskeletal health through prevention of degeneration, tissue repair and tissue regeneration. The HSS Global Innovation Institute was formed in 2016 to realize the potential of new drugs, therapeutics and devices. The HSS Education Institute is the world's leading provider of education on musculoskeletal health, with its online learning platform offering more than 600 courses to more than 21,000 medical professional members worldwide. Through HSS Global Ventures, the institution is collaborating with medical centers and other organizations to advance the quality and value of musculoskeletal care and to make world-class HSS care more widely accessible nationally and internationally. www.hss.edu.
Return to top of page.
| |
|
Mar 23 2020 Fetal Timeline Maternal Timeline News
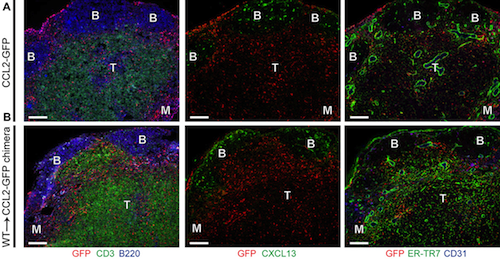 Fibroblastic Reticular Cells (FRCs), are a type of lymph node stromal cell that regulates the survival of antibody-producing plasma cells. The research shows FRCs are a critical source of Chemokine Ligand 2 (CCL2). CCL2 slows expansion of plasma cells. Plasma cells do not generate the CCL2 receptor CCR2; but respond by generating molecules that relay signals from FRCs to plasma cells. This research adds to the role of stromal lymph node cells in fine-tuning our immune responses.
|



