|
|
Developmental Biology - Stem Cells
How Stem Cells Become Organs
Molecular signals dictate which organs embryonic stem cells will become in the first two weeks of embryo development....
In an intricate process guided by so-called "pioneer transcription factors", tightly packed DNA inside each cell is accessed by specialized "pioneer proteins" that activate needed genes. However, until now, it was unclear how these pioneer factors accessed DNA.
Early in each cell, a critical protein known as FoxA2 simultaneously binds to chromosomal proteins and DNA, opening the flood gates for gene activity.
"We now understand that the pioneer factor, FoxA2, grabs chromosomal proteins known as histones, exposing the DNA region," explains Kenneth S. Zaret PhD, the Joseph Leidy Professor in the Department of Cell and Developmental Biology and Director of Penn's Institute for Regenerative Medicine (IRM)."The opening process allows other specialized, regulatory proteins to access DNA in order to activate networks of silent genes leading to the formation of internal organs."
Knowing how regulatory gene proteins work during this early stage can help the field better understand how to control the process of cell development for both (1) clinical research and (2) therapeutic purposes.
Kenneth S. Zaret PhD, Institute for Regenerative Medicine, Department of Cell and Developmental Biology, Perelman School of Medicine, University of Pennsylvania, Philadelphia, PA, USA
For decades, Penn researchers have been pulling back the curtain on this process as they work toward developing new cells for transplantation and tissue repair as treatment for common problems like liver or heart disease.
Zaret's lab previously discovered pioneer factors in 2002 and has been working to better understand their function and role in early embryonic development. In this latest study, the team of researchers, co-led by Makiko Iwafuchi PhD, who performed the work while at Penn and is now at the University of Cincinnati College of Medicine, first used in vitro genetic techniques to investigate the interaction of FoxA with chromosomal proteins at the same time it interacts with DNA. They found that a small region of the FoxA2 protein - just 10 amino acids of the more than 460 - were needed for the protein to make an opening in the chromatin fiber.
The teams translated their findings into a mouse model, deleting the same sequences in mice to see how those changes would affect embryonic development. Removing key amino acid signatures significantly impaired embryonic development, causing deformities in organs - including brains and the heart - which resulted in death in the mice.
"This very small deletion in the protein had a profound effect that mirrored what we had seen in vitro, which surprised us," Zaret's explains. "We originally thought it would be a broadly acting phenomenon that would be hard to pinpoint, but we nailed it down. To see this biochemistry approach, which others were skeptical of, so clearly illuminate a facet of developmental biology - was a real thrill."
Zaret's lab continues to investigate FoxA and other pioneer factors to learn how they may open up the chromatin and interact with chromosomal proteins, similar to FoxA or perhaps in other ways. The current findings serve as a road map.
"Now that we have these results, we are emboldened to investigate diverse other proteins that behave this way. We know that FoxA2 doesn't act alone in turning on the endoderm program to make organs, and we're currently working to better understand how the different factors play a role in that development."
Kenneth S. Zaret PhD.
Abstract
Gene network transitions in embryos and other fate-changing contexts involve combinations of transcription factors. A subset of fate-changing transcription factors act as pioneers; they scan and target nucleosomal DNA and initiate cooperative events that can open the local chromatin. However, a gap has remained in understanding how molecular interactions with the nucleosome contribute to the chromatin-opening phenomenon. Here we identified a short å-helical region, conserved among FOXA pioneer factors, that interacts with core histones and contributes to chromatin opening in vitro. The same domain is involved in chromatin opening in early mouse embryos for normal development. Thus, local opening of chromatin by interactions between pioneer factors and core histones promotes genetic programming.
Authors
Makiko Iwafuchi, Isabel Cuesta, Greg Donahue, Naomi Takenaka, Anna B. Osipovich, Mark A. Magnuson, Heinrich Roder, Steven H. Seeholzer, Pilar Santisteban and Kenneth S. Zaret.
Acknowledgements
The authors thank R. Dunbrack (Fox Chase Cancer Center) for the secondary structure analysis, C. Ducker for the crosslinking studies, S. Hipkens for the mouse embryonic stem cell culture and gene targeting in the Transgenic Mouse/Embryonic Stem Cell Shared Resource, R. McCarthy for data review and the University of Pennsylvania Flow Cytometry and Cell Sorting Facility. M.I. was supported by postdoctoral fellowships from Japan Society for the Promotion of Science Foundation (H26-683), Naito Foundation (RYU10000032), Astellas Foundation for Research on Metabolic Disorders (K0076) and Uehara Memorial Foundation (H24-20124). P.S. was supported by grant nos. SAF2016-75531-R (MICINN/FEDER, UE) and B2017/BMD-3724 (Comunidad de Madrid). The research was supported by NIH grant no. GM36477 to K.S.Z.
Penn co-authors on the study include Greg Donahue and Naomi Takenaka. The study was initiated with Isabel Cuesta and Pilar Santisteban of the Autonomous University of Madrid and included Anna B. Osipovich and Mark A. Magnuson of Vanderbilt University. The study was supported, in part, by the National Institutes of Health (GM36477) and institutional grants.
Penn Medicine is one of the world's leading academic medical centers, dedicated to the related missions of medical education, biomedical research, and excellence in patient care. Penn Medicine consists of the (founded in 1765 as the nation's first medical school) and the University of Pennsylvania Health System, which together form a $7.8 billion enterprise.
The Perelman School of Medicine has been ranked among the top medical schools in the United States for more than 20 years, according to U.S. News & World Report's survey of research-oriented medical schools. The School is consistently among the nation's top recipients of funding from the National Institutes of Health, with $425 million awarded in the 2018 fiscal year.
The University of Pennsylvania Health System's patient care facilities include: the Hospital of the University of Pennsylvania and Penn Presbyterian Medical Center - which are recognized as one of the nation's top "Honor Roll" hospitals by U.S. News & World Report - Chester County Hospital; Lancaster General Health; Penn Medicine Princeton Health; and Pennsylvania Hospital, the nation's first hospital, founded in 1751. Additional facilities and enterprises include Good Shepherd Penn Partners, Penn Home Care and Hospice Services, Lancaster Behavioral Health Hospital, and Princeton House Behavioral Health, among others.
Penn Medicine is powered by a talented and dedicated workforce of more than 40,000 people. The organization also has alliances with top community health systems across both Southeastern Pennsylvania and Southern New Jersey, creating more options for patients no matter where they live.
Penn Medicine is committed to improving lives and health through a variety of community-based programs and activities. In fiscal year 2018, Penn Medicine provided more than $525 million to benefit our community.
Return to top of page.
| |
|
Mar 25 2020 Fetal Timeline Maternal Timeline News
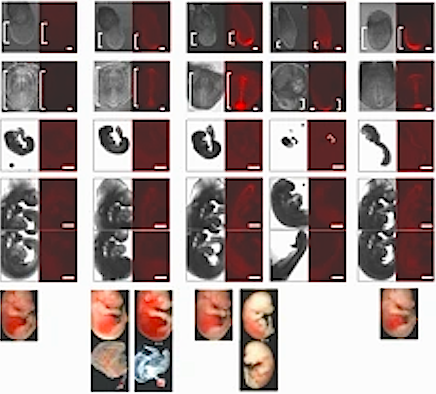
FOXA pioneer factor identified as key to interaction with stem cell histones initiating
gene programming of fetal organs. CREDIT University of Pennsylvania.
|



