|
|
Developmental Biology - Human Body Segmentation Clock
Reconstructing the Human Clock
Using human iPS cells to decipher cell timing at the beginning of life...
From the first division in a fertilized egg, a complex network of proteins and genes push-and-pull on cells constructing the pattern of our organs.
Like the pendulum on a clock, each swing and pulse needs to carefully align, to maintain the rhythm that forms life.
However, much of our understanding of early human development is exceedingly limited, a key reason being our lack of experimental models capable of reproducing these complex biological processes.
Now, researchers led by Kyoto University have reconstituted the human segmentation clock a key focus of embryo developmental research using induced pluripotent stem cells or iPSCs.
"A process called somitogenesis begins about 20 days after fertilization in humans. This is when the embryo develops distinct segments called 'somites' and determines the basic segmented pattern of the body. Somites eventually contribute to the formation of the vertebrae and ribs."
Cantas Alev PhD, Kyoto University, Institute for Advanced Study of Human Biology (ASHBi) and team leader.
Emergence of somites is determined by the segmentation clock, a genetic oscillator that controls and guides the emergence of somites. While segmentation clock genes and their role in development have been studied in mice, chicks, and zebrafish, almost nothing is known about them in humans.
A way of addressing this is reconstruction of the segmentation clock using stem cells. A team consisting of members from ASHBi, Kyoto University's Center for iPS Cell and Research Application, and RIKEN focused on human iPS cells to form a pre-somitic mesoderm precursor cells of somites. Their paper is published in Nature.
"We began by mimicking the signaling pathways active during early development. Applying our knowledge of embryology, we succeeded in generating a culture of pre-somitic mesoderm, or PSM, along with its progeny cells. Studying genes that were expressed in a rhythmic pattern showed not only that they oscillated within a period of five hours, but also revealed novel genetic components of the 'segmentation clock' we were looking for."
Cantas Alev PhD, Department of Cell Growth and Differentiation, Center for iPS Cell Research and Application (CiRA), Kyoto University, Kyoto, Japan.
In addition to the oscillation of genes, the team replicated a second hallmark of the segmentation clock, a wave of expression. Using gene-editing technology, they then assessed the function of key genes related to spine deformation.
As expected, mutations in these genes dramatically altered aspects of the segmentation clock including synchronization and oscillation. They went further and generated iPS cells from patients with aforementioned genetic defects, identified the mutations involved, and corrected them.
The study demonstrates how elegantly iPS cells can be used to recapitulate distinct aspects of human embryonic development and other complex biological processes.
"Like many developmental biologists I am fascinated by embryos and embryonic development. The elegance and beauty of how complex organs and tissues are formed from very simple initial structures is astounding. I hope to reconstruct and analyze many other aspects of embryonic development, and expand our still limited understanding of human and non-human development."
Cantas Alev PhD
Abstract
Pluripotent stem cells are increasingly used to model different aspects of embryogenesis and organ formation1. Despite recent advances in in vitro induction of major mesodermal lineages and cell types2,3, experimental model systems that can recapitulate more complex features of human mesoderm development and patterning are largely missing. Here we used induced pluripotent stem cells for the stepwise in vitro induction of presomitic mesoderm and its derivatives to model distinct aspects of human somitogenesis. We focused initially on modelling the human segmentation clock, a major biological concept believed to underlie the rhythmic and controlled emergence of somites, which give rise to the segmental pattern of the vertebrate axial skeleton. We observed oscillatory expression of core segmentation clock genes, including HES7 and DKK1, determined the period of the human segmentation clock to be around five hours, and demonstrated the presence of dynamic travelling-wave-like gene expression in in vitro-induced human presomitic mesoderm. Furthermore, we identified and compared oscillatory genes in human and mouse presomitic mesoderm derived from pluripotent stem cells, which revealed species-specific and shared molecular components and pathways associated with the putative mouse and human segmentation clocks. Using CRISPRCas9-based genome editing technology, we then targeted genes for which mutations in patients with segmentation defects of the vertebrae, such as spondylocostal dysostosis, have been reported (HES7, LFNG, DLL3 and MESP2). Subsequent analysis of patient-like and patient-derived induced pluripotent stem cells revealed gene-specific alterations in oscillation, synchronization or differentiation properties. Our findings provide insights into the human segmentation clock as well as diseases associated with human axial skeletogenesis.
Authors
Mitsuhiro Matsuda, Yoshihiro Yamanaka, Maya Uemura, Mitsujiro Osawa, Megumu K. Saito, Ayako Nagahashi, Megumi Nishio, Long Guo, Shiro Ikegawa, Satoko Sakurai, Shunsuke Kihara, Thomas L. Maurissen, Michiko Nakamura, Tomoko Matsumoto, Hiroyuki Yoshitomi, Makoto Ikeya, Noriaki Kawakami, Takuya Yamamoto, Knut Woltjen, Miki Ebisuya, Junya Toguchida and Cantas Alev.
Acknowledgements
The authors thank B. McIntyre and P. ONeill for critical reading of the manuscript; K. Mitsunaga for help with FACS analysis; Y. Ashida for help with development of human spheroid PSM-induction protocol; H. Hayashi for help with development of mouse PSM protocol; J. Asahira for help with RNA-seq experiments; A. Yamashita for help with 3D chondrogenic induction experiments; M. Shibata and T. Nakajima for help with development of one-step PSM-induction protocol; M. Ohno and S. Nishimura for help with iPS cell quality control and validation; members of the Kageyama laboratory, K. Yoshioka-Kobayashi and A. Isomura for help with Hilbert transformation and M. Matsumiya for help with removing spike noise from images; the CiRA Genome Evaluation Group, in particular H. Dohi, F. Kitaoka, M. Nomura, T. Takahashi, M. Umekage and N. Takasu for performing SNP array analysis. This work was supported by the CiRA Fellowship Program of Challenge to C.A.; Naito Foundation Research Grant to C.A.; Grant-in-Aid for Challenging Exploratory Research (KAKENHI Number 16K15664) to C.A.; Grant-in-Aid for Scientific Research on Innovative Areas (KAKENHI Number 17H05777) to M.M.; Takeda Science Foundation Grant to M.E.; Japan Agency for Medical Research and Development (AMED) Grants Number 12103610 and 17935423 to M.K.S. for iPS cell generation and qualification, grant number JP19bm0804001 to K.W. for iPS cell gene editing and grant numbers JP18ek0109212 and 18ek0109280 to S.I. for genomic and exome studies of spondylocostal dysostosis, respectively; the Core Center for iPS Cell Research (AMED) to T.Y., K.W. and J.T. and the Acceleration Program for Intractable Disease Research Using Disease Specific iPS Cells (AMED) to K.W., J.T. and M.K.S.; the Kyoto University Hakubi Project to K.W.; the Cooperative Research Program (Joint Usage/Research Center Program) of the Institute for Frontier Life and Medical Sciences, Kyoto University to J.T., L.G and S.I.. ASHBi is supported by the World Premier International Research Center Initiative (WPI), MEXT, Japan.
About Kyoto University
Kyoto University is one of Japan and Asia's premier research institutions, founded in 1897 and responsible for producing numerous Nobel laureates and winners of other prestigious international prizes. A broad curriculum across the arts and sciences at both undergraduate and graduate levels is complemented by numerous research centers, as well as facilities and offices around Japan and the world. For more information please see: http://www.kyoto-u.ac.jp/en
Return to top of page.
| |
|
Apr 17 2020 Fetal Timeline Maternal Timeline News
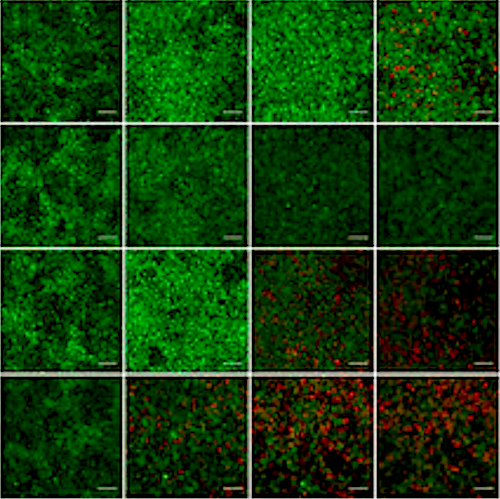 ASHBi research has reconstructed oscillation patterns in the somatic cell segmentation clock using human iPS cells in orer to analyze those key genes involved and correct errors. CREDIT Kyoto University: Cantas Alev & Misaki Ouchida. Access via: Research Gate
|



