|
|
Developmental Biology - Pregnant Immune System
The Pregnancy Immune System
A mother's immune system works differently between her first and her later pregnancies...
Among the holy quests of medical challenges doctors and research scientists strive to vanquish are miscarriage, stillbirth and preterm birth.
Sing Sing Way, MD, PhD and director and principal investigator of the Center for Inflammation and Tolerance, Cincinnati Children's Hospital Medical Center, is part of a small army of scientists, physicians and others looking for answers to basic pregnancy questions with enormous health and scientific implications. Their goal is to devise improved therapies to stem the tide of what remains a severe and entrenched public health crisis.
Way's researchers focused on how the immune system changes during pregnancy. The scientists used animal pregnancy models to evaluate differences in human pregnancy outcomes. This included evaluating maternal age, genetic diversity between parents, and prior pregnancies.
A mother's immune system is altered during pregnancy to prevent fetal rejection by her body. It is a delicate immune balance. Disruption could lead to a miscarriage or multiple miscarriages. Unfortunately, that balance is too often disrupted by environment or physiological change leading to pregnancy complications.
Way's team now adds an important wrinkle, reporting in Cell Reports that a woman's immune system behaves very differently between a first and second pregnancy. The researchers hope this knowledge can lead to more personalized and more effective therapeutic pregnancy strategies.
"First and subsequent pregnancies work very differently. Understanding these differences can lead to improved therapies targeting unique immunological differences occurring in first and later pregnancies."
Sing Sing Way, MD, PhD, Division of Infectious Diseases, Center for Inflammation and Tolerance, Cincinnati Childrens Hospital Medical Center, University of Cincinnati College of Medicine, Cincinnati, Ohio, USA.
The study shows immune pathways promoting a healthy first pregnancy are not the same pathways that promote later pregnancies.
Pregnancy causes physiological exposure, and often re-exposure, to foreign fetal alloantigens expressed by the developing fetus.
These fetal alloantigens interact directly with the mother's immune system with highly variable consequences.
Thus researchers found evidence of both alloimmunization (where the immune system attacks) as well as when the immune system expands tolerance and does not attack. This data shows that pregnancy primes the accumulation of fetal-specific maternal CD8+ T cells, that give mothers their ability to 'remember' their babies immune system. These cells persist as active memory pools after a mother gives birth.
The function of memory T cell PD-1 and LAG-3 proteins, reminds cells to again be tolerant of the developing fetus during subsequent pregnancies. But according to research associate and study first author Jeremy Kinder, molecular disruption can neutralize the function of proteins PD-1 and LAG-3, activating fetal CD8+ T cells and cause miscarriage in subsequent pregnancies.
Abstract Summary
Highlights
Pregnancy primes CD8+ T cells with fetal specificity that persists after pregnancy
Memory CD8+ T cells are cytolytic and primed for effector cytokine production
Fetal-specific CD8+ T cells are functionally exhausted during secondary pregnancy
PD-1 and LAG-3 suppress CD8+ T cells selectively with fetal antigen re-stimulation
Summary
Pregnancy necessitates physiological exposure, and often re-exposure, to foreign fetal alloantigens. The consequences after pregnancy are highly varied, with evidence of both alloimmunization and expanded tolerance phenotypes. We show that pregnancy primes the accumulation of fetal-specific maternal CD8+ T cells and their persistence as an activated memory pool after parturition. Cytolysis and the potential for robust secondary expansion occurs with antigen re-encounter in non-reproductive contexts. Comparatively, CD8+ T cell functional exhaustion associated with increased PD-1 and LAG-3 expression occurs with fetal antigen re-stimulation during subsequent pregnancy. PD-L1/LAG-3 neutralization unleashes the activation of fetal-specific CD8+ T cells, causing fetal wastage selectively during secondary but not primary pregnancy. Thus, CD8+ T cells with fetal alloantigen specificity persist in mothers after pregnancy, and protection against fetal wastage in subsequent pregnancies is maintained by their unique susceptibility to functional exhaustion. Together, distinct mechanisms whereby fetal tolerance is maintained during primary compared with subsequent pregnancies are demonstrated.
Authors
Jeremy M. Kinder,Lucien H. Turner, Ina A. Stelzer, Hilary Miller-Handley, Ashley Burg, Tzu-Yu Shao, Giang Pham and Sing Sing Way.
Acknowledgements
The researchers are indebted to Drs. Theresa Alenghat, Hitesh Deshmukh, and Chandrashekhar Pasare, and Joseph Qualls for helpful suggestions.
Funding support for the study came in part from the National Institutes of Health (R01AI120202, R01AI124657 and DP1AI131080); the HHMI Faculty Scholar's program , the March of Dimes Ohio Collaborative for Prematurity Research, and a Burroughs Wellcome Fund Investigator in the Pathogenesis Award.
Return to top of page.
|
|
Jul 3 2020 Fetal Timeline Maternal Timeline News
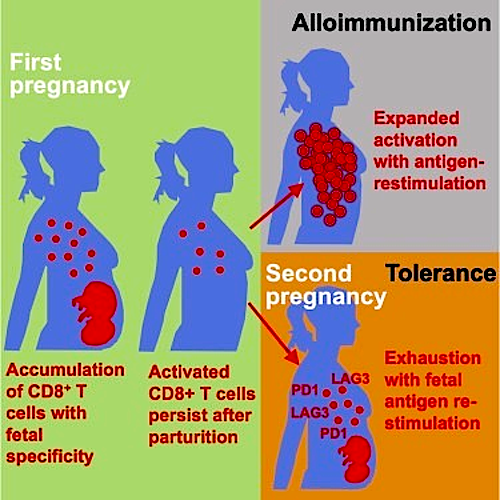 A woman's immune system behaves very differently between a first and later pregnancies. CREDIT: Medical University of South Carolina (MUSC), Hollings Cancer Center (HCC)
|
|

