|

CLICK ON weeks 0 - 40 and follow along every 2 weeks of fetal development
|
||||||||||||||||||||||||||||
|
Developmental Biology - Epithelial Cells Sheets of Epithelial Cells Line Every Organ This model takes into account all mechanical forces between neighboring cells, leading to a formula for the total mechanical energy in an epithelial sheet. Measurment of the function of only a few parameters, including V and n, as well as the in-plane density produces a quantity called sharpness, distinguished by its location between situations where basal and/or apical surfaces are present or not. (A vanished apical surface implies basal extrusion and vice versa.) By studying how energy changes based on these few parameters, researchers were able to determine the mechanics of an epithelial sheet. The key finding of Okuda and Fujimoto is that this simple layer system is inherently unstable! They also found many agreements between their Model outcomes and observations in living systems. Such as in the occurrence of different epithelial geometries (e.g. rosette or pseudostratified structures). The authors recognize that their model has limitations, for example it assumes the whole sheet and individual cell surfaces are not curved but flat. "Despite its limitations, this model provides a guide to understanding the wide range of epithelial physiology that occurs in morphogenesis, homeostasis, and carcinogenesis." About Epithelial cells Epithelial tissue, one of four kinds of human (or animal) tissue, is located on the outer surfaces of organs and blood vessels in the human body, and on the inner surfaces of 'hollow spaces' in various internal organs. A typical example is the outer layer of the skin, called the epidermis. Epithelial tissue consists of epithelial cells; it can be just one layer of epithelial cells (simple epithelium), or two or more (layered or stratified epithelium). Satoru Okuda and Koichi Fujimoto from Kanazawa University have now modeled a simple epithelium as an arrangement of polyhedra in order to study its mechanical properties and specifically the process of epithelial cell extrusion. About Cell extrusion In epithelial tissue, cell extrusions happen -- the processes whereby epithelial cells are 'pushed out' of the epithelium. Cell extrusion is an important biological process, regulating for example the removal of apoptotic (dead) cells, tissue growth and the response to cancer. Okuda and Fujimoto looked at a simple epithelium from a mechanical point of view. Modeling the epithelium as a layer of interconnected polyhedra, they found that cell extrusion -- whereby the top or bottom surface of a polyhedron shrinks to a point and then vanishes -- can be considered a purely mechanical property. An inherent instability, present in homogeneous sheets, can lead to cells being extruded due to small changes in density or topology. Abstract Highlights In cell extrusion, a cell embedded in an epithelial monolayer loses its apical or basal surface and is subsequently squeezed out of the monolayer by neighboring cells. Cell extrusions occur during apoptosis, epithelial-mesenchymal transition, or precancerous cell invasion. They play important roles in embryogenesis, homeostasis, carcinogenesis, and many other biological processes. Although many of the molecular factors involved in cell extrusion are known, little is known about the mechanical basis of cell extrusion. We used a three-dimensional (3D) vertex model to investigate the mechanical stability of cells arranged in a monolayer with 3D foam geometry. We found that when the cells composing the monolayer have homogeneous mechanical properties, cells are extruded from the monolayer when the symmetry of the 3D geometry is broken because of an increase in cell density or a decrease in the number of topological neighbors around single cells. Those results suggest that mechanical instability inherent in the 3D foam geometry of epithelial monolayers is sufficient to drive epithelial cell extrusion. In the situation in which cells in the monolayer actively generate contractile or adhesive forces under the control of intrinsic genetic programs, the forces act to break the symmetry of the monolayer, leading to cell extrusion that is directed to the apical or basal side of the monolayer by the balance of contractile and adhesive forces on the apical and basal sides. Although our analyses are based on a simple mechanical model, our results are in accordance with observations of epithelial monolayers in vivo and consistently explain cell extrusions under a wide range of physiological and pathophysiological conditions. Our results illustrate the importance of a mechanical understanding of cell extrusion and provide a basis by which to link molecular regulation to physical processes. Significance Epithelial cell extrusion is important for biological processes such as embryogenesis, homeostasis, and carcinogenesis. Various molecular factors, such as cancer genes and their products, have been reported as key drivers of epithelial extrusion; however, little is known about how these factors are linked to the mechanical process. A simple mathematical model based on mechanics can consistently explain cell extrusions under a wide range of physiological and pathophysiological conditions. The model shows that cells can be extruded from homogeneous sheets, owing to the inherent mechanical instability of the 3D foam geometry of the epithelial monolayer. When the cells generate active forces, the forces act to enhance the instability and direct extrusion to either the apical or basal side of the monolayer. Authors Satoru Okuda, WPI Nano Life Science Institute, Kanazawa University, Kakuma-cho, Kanazawa, Japan; and Koichi Fujimoto, Department of Biological Sciences, Osaka University, Machikaneyama-cho, Toyonaka, Japan. Acknowledgements The authors thank thank Dr. Tetsuya Hiraiwa at University of Tokyo, Dr. Romain Levayer at Institut Pasteur, Dr. Yosuke Ogura at RIKEN Center for Biosystems Dynamics Research, Dr. Katsuyoshi Matsushita at Osaka University, and Mr. Koki Nunota at Kanazawa University for discussions. S.O. thanks his colleagues in the laboratory of Prof. Mototsugu Eiraku at Kyoto University for discussions. This work was supported by the Japan Science and Technology Agency (JST), CREST Grant No. JPMJCR1921 (S.O.), PRESTO Grant No. JPMJPR16F3 (S.O.); the Japan Society for the Promotion of Science (JSPS), KAKENHI Grants No. 17KT0021 (S.O.), 17H02939 (S.O.), 17H05619 (K.F.), 19H04777 (S.O.); the Uehara Memorial Foundation, Japan (S.O.); and the World Premier International Research Center Initiative, Ministry of Education, Culture, Sports, Science and Technology (MEXT), Japan (S.O.). Return to top of page.
|
| Jul 21 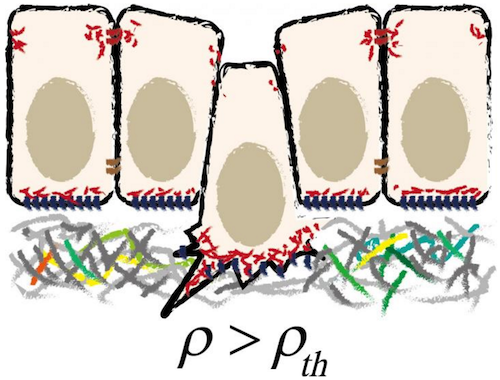 Schematic illustration of cell extrusion from epithelial tissue. CREDIT KANAZAWA UNIVERSITY
| |||||||||||||||||||||||||||

