|

CLICK ON weeks 0 - 40 and follow along every 2 weeks of fetal development
|
||||||||||||||||||||||||||||
|
Developmental Biology - Protein Regulation How Cells Keep Growing Even When Under Attack That connection led researchers to determine damaged proteins arise when stress blocks Lon. Highlights Loss of Lon increases dNTP pools to permit loss of normally essential dCTP deaminase Stabilization of CcrM increases RNR expression and protects against hydroxyurea Misfolded proteins can competitively inhibit CcrM degradation by the Lon protease Stabilizing transcription factors by titration of Lon promotes stress responses Summary During proteotoxic stress, bacteria maintain critical processes like DNA replication while removing misfolded proteins, which are degraded by the Lon protease. Here, we show that in Caulobacter crescentus Lon controls deoxyribonucleoside triphosphate (dNTP) pools during stress through degradation of the transcription factor CcrM. Elevated dNTP/nucleotide triphosphate (NTP) ratios in Delta Lon cells protects them from deletion of otherwise essential deoxythymidine triphosphate (dTTP)-producing pathways and shields them from hydroxyurea-induced loss of dNTPs. Increased dNTP production in Delta Lon results from higher expression of ribonucleotide reductase driven by increased CcrM. We show that misfolded proteins can stabilize CcrM by competing for limited protease and that Lon-dependent control of dNTPs improves fitness during protein misfolding conditions. We propose that linking dNTP production with availability of Lon allows Caulobacter to maintain replication capacity when misfolded protein burden increases, such as during rapid growth. Because Lon recognizes misfolded proteins regardless of the stress, this mechanism allows for response to a variety of unanticipated conditions. Authors Rilee D. Zeinert, Hamid Baniasadi, Benjamin P. Tu and PeterChien. Acknowledgements This work was funded by the National Institute of General Medical Sciences in the form of a MIRA grant to Chien and the Chemistry-Biology Training Program, which also supported Zeinert. The MIRA program does not fund individual projects, but broad programs of basic discovery research, to encourage researchers to propose more long-term, innovative, creative projects and to worry less about short-term goals and results. D.M.G. and M.Z.-G. are grateful to Caltech for start-up funding. Work in the M.Z.-G. laboratory is also funded by grants from the Wellcome Trust (207415/Z/17/Z); Open Philanthropy; the Weston Havens Foundation; and the Curci Foundation. B.A.T.W. is supported by the Gates Cambridge Trust. The authors declare they have no competing interests. Return to top of page.
|
| Aug 10 2020 Fetal Timeline Maternal Timeline News 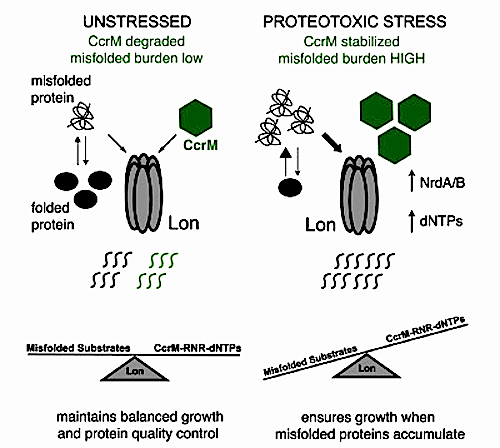 CREDIT The Authors.
| |||||||||||||||||||||||||||

