|
|
Developmental Biology - Krabbe Disease
Towards a Treatment for Krabbe Disease
In one of 100,000 births, a mutation in the GALC gene causes an incurable, fatal disorder known as infantile Krabbe disease. Most toddlers with the condition die within 2 years...
Krabbe disease, or globoid cell leukodystrophy, is inherited and typically kills children before their second birthday. It has no cure. Now, a University of Pennsylvania study on a canine model offers hope for an effective gene therapy to compensate for the mutated galactosylceramidase gene.
A parallel condition also affects dogs. Puppies typically show symptoms of the disease at six weeks old and succumb within a few months. A study in the Journal of Clinical Investigation (JCI), led by Charles H. Vite BS DVM PhD, Professor of Neurology, University of Pennsylvania School of Veterinary Medicine, found gene therapy for Krabbe disease in dogs had the most enduring impact.
Dogs that receive treatment have lived to 4 years of age and beyond with no significant symptoms. This research highlights the potential for a similar treatment approach in children.
"This disease has no good therapy," explains Vite, senior author on the paper. "We've been working with dogs since the 1990s. However, the shift to a new gene therapy vector created a positive effect on their nervous system."
Krabbe disease is one of a group of lysosomal storage diseases characterized by a buildup of lipids in lysosome organelles located within cells. Normally, the GALC gene encodes an enzyme that breaks down lipids in the body. In Krabbe disease, the mutated GALC causes lipids to build up, resulting in deformed growth of the lipid-containing coating of nerve cells, and in the myelin sheath leading to impaired nerve cell signaling. As a result, children with Krabbe disease experience progressive neurological symptoms, including blindness, deafness, and paralysis.
Symptoms of Lysosomal Storage Diseases
Symptoms vary depending on the type of lysosomal storage disease and one or more may occur:
Delay in Intellectual and Physical Development
Seizures
Facial and other Bone deformities
Joint Stiffness, Pain
Difficulty Breathing
Vision and Hearing Problems
Anemia, Nosebleeds, Easy Bleeding/Bruising
Swollen Abdomen from Enlarged Spleen or Liver
Mental Retardation
Behavioral problems: Aggression & Hyperactivity
Although a bone marrow transplant within the first month of life can prevent symptoms from begining in about 30% of infants, the procedure is exceedingly risky. Vite: "A new treatment is really needed."
Krabbe disease was one of the first pediatric genetic diseases for which a parallel inherited disorder was found in canines. Dogs with the condition are part of Penn Vet's Referral Center for Animal Models of Human Genetic Disease, allowing for investigation of possible human therapies.
To blunt the effects of the disease, researchers knew getting the GALC gene into the brain was crucial. They found the AAV9 vector, used effectively in experimental gene therapies for other neurological diseases, to be the best candidate for FDA approval.
The site of delivery was also important. "We decided we would inject into the spinal fluid via the back of the head, which is the most effective means of getting into the brain," explained Vite.
The researchers used both a high and low dose of gene therapy, administering it to dogs that were two weeks old, before symptoms appear, or six weeks old, after neurological symptoms had begun to emerge.
Vite and colleagues worried that simply delivering a normal copy of GALC would not alleviate Krabbe symptoms, due to buildup of toxic psychosine - left by metabolism of the mutated GALC enzyme. But, they became very excited by the dramatic results they saw.
Dogs receiving high dose gene therapy before the onset of symptoms not only had healthy myelination in their brains, they maintained myelination in their peripheral nervous systems. Vite: "That was a huge surprise! Injecting gene therapy into the spinal fluid positively affected both the central and peripheral nervous systems."
Gene therapy dogs have lived symptom-free for up to four years. Even dogs treated after showing symptoms lived significantly longer than those not receiving therapy. However, lower doses of gene therapy, resulted in an intermediate form of the disease, underscoring the importance of correct dosing when translating these findings to children.
Allison Bradbury PhD, lead author of the JCI paper, is now assistant professor in the Department of Pediatrics, Ohio State University; and a principal investigator in the Center for Gene Therapy, Abigail Wexner Research Institute at Nationwide Children's Hospital.
and an investigator at Nationwide Children's Hospital where she continues to investigate how gene therapy lowers psychosine levels.
Vite's group, continues to research how biological differences and size influence dogs and children in their response to gene therapy, and to identify an effective dose for both. Given the beneficial effects of gene therapy, as observed in Krabbe affected peripheral nerves, he is hopeful this approach will work for other peripheral nervous system diseases.
"Our hope is to use the model as a method to understand the mechanisms at work in peripheral nerves and then target peripheral neuropathies."
Charles Vite PhD, Professor, Neurology, University of Pennsylvania School of Veterinary Medicine.
Abstract
Globoid cell leukodystrophy (GLD; Krabbe disease) is a progressive, incurable neurodegenerative disease caused by deficient activity of the hydrolytic enzyme galactosylceramidase (GALC). The ensuing cytotoxic accumulation of psychosine results in diffuse central and peripheral nervous system (CNS, PNS) demyelination. Presymptomatic hematopoietic stem cell transplantation (HSCT) is the only treatment for infantile-onset GLD; however, clinical outcomes of HSCT recipients often remain poor, and procedure-related morbidity is high. There are no effective therapies for symptomatic patients. Herein, we demonstrate in the naturally occurring canine model of GLD that presymptomatic monotherapy with intrathecal AAV9 encoding canine GALC administered into the cisterna magna increased GALC enzyme activity, normalized psychosine concentration, improved myelination, and attenuated inflammation in both the CNS and PNS. Moreover, AAV-mediated therapy successfully prevented clinical neurological dysfunction, allowing treated dogs to live beyond 2.5 years of age, more than 7 times longer than untreated dogs. Furthermore, we found that a 5-fold lower dose resulted in an attenuated form of disease, indicating that sufficient dosing is critical. Finally, postsymptomatic therapy with high-dose AAV9 also significantly extended lifespan, signifying a treatment option for patients for whom HSCT is not applicable. If translatable to patients, these findings would improve the outcomes of patients treated either pre- or postsymptomatically.
Authors
Allison M. Bradbury, Jessica H. Bagel, Duc Nguyen, Erik A. Lykken, Jill Pesayco Salvador, Xuntian Jiang, Gary P. Swain, Charles A. Assenmacher, Ian J. Hendricks, Keiko Miyadera, Rebecka S. Hess, Arielle Ostrager, Patricia ODonnell, Mark S. Sands, Daniel S. Ory, G. Diane Shelton, Ernesto R. Bongarzone, Steven J. Gray and Charles H. Vite.
Acknowledgements
FUNDER
National Institutes of Health.
Return to top of page.
| |
|
Aug 27 2020 Fetal Timeline Maternal Timeline News
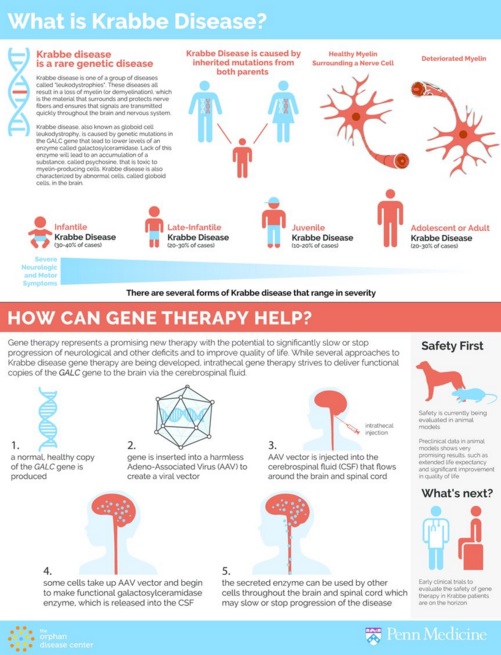 Krabbe disease is one of a group of lysosomal storage diseases where a buildup of lipids (molecules that are the building blocks, structure and function of cells) interfers with signals sent from cell to cell. It affects one in 100,000, and can be fatal before the age of 2. CREDIT Penn Medicine.
|



