|
|
Developmental Biology - Flu Virus in Pregnancy
A Flu-Induced Vascular Storm in Pregnancy
New research explains how the flu virus can lead to life-threatening complications in pregnancy...
A pre-clinical study has overturned current scientific thinking on why flu infections affect pregnant women and their babies so severely. The findings could also help explain the biology behind COVID-19 spread from the lungs.
Research in animal models (mice), reveals that during pregnancy flu spreads from the lungs via blood vessels in our circulatory system, triggering damaging hyperactive immune responses. Led by Royal Melbourne Institute of Technology and Melbourne Technical College (RMIT) in collaboration with researchers and clinicians from RMIT Ireland and Australia, the work is published in PNAS.
According to lead author Stella Liong PhD, this new research suggests the vascular system is at the heart of the potentially devastating complications caused by influenza during pregnancy.
"We've known for a long time that flu can cause serious maternal and fetal complications, but how this happens has not been clearly understood," Liong, a Vice-Chancellor's Postdoctoral Fellow at RMIT, School of Health and Biomedical Sciences, Royal Melbourne Institute of Technology University, Bundoora, Australia explains: "Conventional thinking has blamed the suppressed immune system that occurs in pregnancy. But what we see is the opposite effect, flu infection leads to a drastically heightened immune response.
Inflammation we found in the circulatory system is so overwhelming, it's like a vascular storm wreaking havoc throughout the body. We need further research to clinically validate our findings but the discovery of this new mechanism is a crucial step towards the development of flu therapies designed specifically for pregnant women."
"The discovery of an influenza-induced 'vascular storm' is one of the most significant developments in inflammatory infectious diseases over the last 30 years with significant implications for other viral infections, including COVID-19."
John O'Leary PhD, Professor, Discipline of Histopathology, School of Medicine, Trinity Translational Medicine Institute, Trinity College Dublin, Ireland.
Influenza is not directly passed from mother to baby, but its potentially devastating effect on the mother is closely connected to complications suffered by the baby. Pregnant women who develop influenza are at higher risk of hospitalisation with pneumonia and other complications, while babies of mothers severely affected by flu are at increased risk of fetal growth restriction, miscarriage and preterm births.
Scientists have previously thought the reason flu has such serious health impacts is because the immune system is suppressed during pregnancy to enable the fetus to thrive making it harder for the mother's body to fight her own infections. However, the new research on Influenza A shows the virus behaves very differently in the bodies of pregnant and non-pregnant mice.
In non-pregnant mice, the flu infection remains localised to the lungs. But in pregnant mice, the virus spreads into the circulatory system via the blood vessels. This leads to intense inflammation that drastically affects the function of large blood vessels, severely impacting health of the mother and can restrict blood flow to the growing fetus.
Flu-Induced Vascular Storm
In the new study, researchers found pregnant mice with flu had severe inflammation in the large blood vessels and the aorta, the major conduit artery from the heart. While a healthy blood vessel dilates 90-100% to let blood flow freely, the flu-infected blood vessels functioned at only 20-30% of capacity.
"Even a small change in the diameter of a blood vessel could have profound changes to blood flow."
Stavros Selemidis PhD, Associate Professor, School of Health and Biomedical Sciences, Royal Melbourne Institute of Technology University, Bundoora, Australia and lead investigator.
"We found a dramatic difference in these inflamed blood vessels, which can seriously affect how much blood makes it to the placenta and all the organs that help support the growing baby," says Selemidis.
"We've known that flu infection in pregnancy results in an increased risk of babies being smaller and suffering oxygen starvation. Our research shows the critical role that the vascular system could be playing in this, with inflammation in the blood vessels reducing blood flow and nutrient transfer from mom to baby."
While the researchers did not directly measure blood flow, the study found an increase in biomarkers for oxygen starvation in the fetuses of the flu-infected mice.
Why Pregnancy Makes a Difference
During pregnancy, the placenta secretes proteins and releases fetal DNA into the mother's blood, which can cause her underlying inflammation. The new study suggests an influenza infection may tip that underlying inflammation in the mother's body into a full-blown systemic inflammatory event.
Selemidis adds that the research also revealed a new connection to pre-eclampsia, a dangerous pregnancy complication characterised by high blood pressure.
"We found the same protein that is elevated in pre-eclampsia is also significantly elevated with flu. While it will take further research to unpack this link, it could mean drugs targeting vascular inflammation that are currently being tested could potentially be repurposed in the future for flu infection in pregnancy."
Stavros Selemidis PhD.
Coronavirus connection,
Liong believes the research also has implications for our understanding of how the COVID-19 virus may be affecting the vascular system.
"Flu and coronavirus are different, but there are parallels. We do know that COVID-19 causes vascular dysfunction - which can lead to strokes and other cardiovascular problems. Our pregnancy studies offer new insights into the fundamental biology of how respiratory viruses can also drive dysfunction in the vascular system. This could be valuable knowledge for those scientists working directly on treatments and vaccines for COVID-19."
Stavros Selemidis PhD and lead investigator.
Significance/Abstract
Significance
Influenza infection during pregnancy is associated with increased maternal and perinatal complications. Here, we show that, during pregnancy, influenza infection leads to viral dissemination into the aorta, resulting in a peripheral vascular storm characterized by enhanced inflammatory mediators; the influx of Ly6C monocytes, neutrophils, and T cells; and impaired vascular function. The ensuing vascular storm induced hypoxia in the placenta and fetal brain and caused an increase in circulating cell free fetal DNA and soluble Flt1 release. We demonstrate that vascular dysfunction occurs in response to viral infection during pregnancy, which may explain the high rates of morbidity and mortality in pregnant dams, as well as the downstream perinatal complications associated with influenza infection.
Abstract
Influenza A virus (IAV) infection during pregnancy causes severe maternal and perinatal complications, despite a lack of vertical transmission of IAV across the placenta. Here, we demonstrate a significant alteration in the maternal vascular landscape that underpins the maternal and downstream fetal pathology to IAV infection in mice. In IAV infection of nonpregnant mice, the local lung inflammatory response was contained to the lungs and was self-resolving, whereas in pregnant mice, virus dissemination to major maternal blood vessels, including the aorta, resulted in a peripheral "vascular storm," with elevated proinflammatory and antiviral mediators and the influx of Ly6Clow and Ly6Chigh monocytes, plus neutrophils and T cells. This vascular storm was associated with elevated levels of the adhesion molecules ICAM and VCAM and the pattern-recognition receptors TLR7 and TLR9 in the vascular wall, resulting in profound vascular dysfunction. The sequalae of this IAV-driven vascular storm included placental growth retardation and intrauterine growth restriction, evidence of placental and fetal brain hypoxia, and increased circulating cell free fetal DNA and soluble Flt1. In contrast, IAV infection in nonpregnant mice caused no obvious alterations in endothelial function or vascular inflammation. Therefore, IAV infection during pregnancy drives a significant systemic vascular alteration in pregnant dams, which likely suppresses critical blood flow to the placenta and fetus. This study in mice provides a fundamental mechanistic insight and a paradigm into how an immune response to a respiratory virus, such as IAV, is likely to specifically drive maternal and fetal pathologies during pregnancy.
Authors
Stella Liong, Osezua Oseghale, Eunice E. To, Kurt Brassington, Jonathan R. Erlich, Raymond Luong, Felicia Liong, Robert Brooks, Cara Martin, Sharon OToole, Antony Vinh, Luke A. J. ONeill, Steven Bozinovski, Ross Vlahos, Paris C. Papagianis, John J. OLeary, Doug A. Brooks, and Stavros Selemidis.
AcknowledgementsScience
The new study is the culmination of over 10 years' work by researchers in the School of Health and Biomedical Sciences at RMIT, leading a global collaboration.
The research was supported by an Australian Research Council (ARC) Future Fellowship and funding from the National Health and Medical Research Council of Australia (NHMRC).
'Influenza A virus causes maternal and foetal pathology via innate and adaptive vascular inflammation in mice', with collaborators from University of South Australia, Trinity College Dublin, La Trobe University and Monash University.
Return to top of page.
| |
|
Sep 23 2020 Fetal Timeline Maternal Timeline News
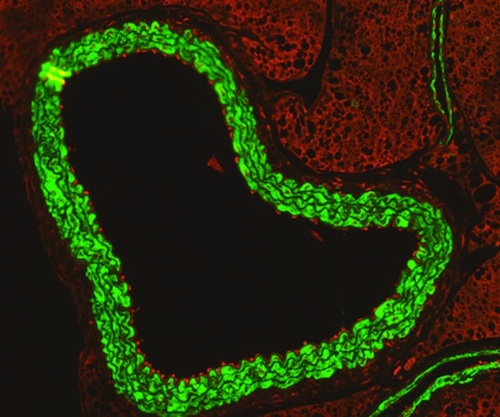 Immunofluorescence image of Activated Immune Cells (RED) 'sticking' to insides of Aorta (GREEN) during FLU Infection in pregnancy. CREDIT RMIT College, Melborne, Australia.
|



