|
|
|
Home | Pregnancy Timeline | News Alerts |News Archive Jul 17, 2015
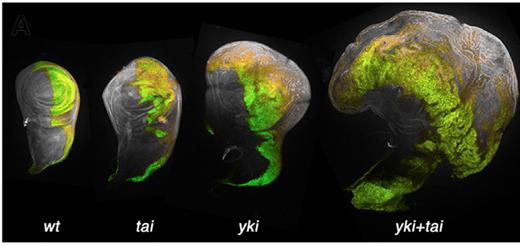
Fruitfly proteins: Taiman (tai) and Yorkie (yki) combine to affect the Hippo pathway
driving abnormal growth in fly wing imaginal discs. Note massive expansion with Tai + Yki.
Image credit: Zhang et al. Developmental Cell 2015
|
|
|
|
|
|
Hippo dances to hormones
Although fruit flies don't develop cancer, stem cell researchers have been learning a great deal from them. In particular, from mutant flies with overgrown organs that resemble hippopotamuses.
A fly gene called Hippo and its gene relative in mammals, normally work to block cell over growth and limit organ size. But mutations in Hippo or genes linked to it - all together called the Hippo pathway - result in cell overgrowth which distorts various tissues into hippopotamus sized bulges in the fly.
Researchers at Emory University School of Medicine have found that the abnormal growth induced by disruption of the Hippo pathway depends on genes which then respond to the steroid hormone ecdysone. "Ecdysone is, to some degree, the fly version of estrogen," says senior author Ken Moberg PhD, associate professor of cell biology at Emory.
In fly larvae, ecdysone triggers metamorphosis,
in which adult wings and eyes emerge from small, circular compartments called imaginal discs. Ecdysone has a chemical structure like that of estrogen, testosterone and steroid hormones found in humans. Although ecdysone is not sex-specific, it acts the same as other steroid hormones, diffusing into cells and binding to proteins that bind DNA and regulate gene activity.
The research team discovered that when the Hippo pathway's control is broken, it results in excess growth of these imaginal discs, depending on which proteins were triggered by the ecdysone.
Genes activated by the combination of Hippo and ecdysone signals from the imaginal discs, can include genes typically only turned on in the germline (sex) stem cells of fly reproductive organs. In fact, researchers found the Hippo pathway requires ecdysone in order to activate germline stem cells. So, the researchers concentrated on a protein in flies called Yorkie, which is usually restrained "upstream" of the Hippo pathway, but when unleashed, travels into the cell nucleus and turns on growth-related genes.
"We found that [the protein] Yorkie does not just engage a developmental growth program in disc tumors. It is capable of turning on an ectopic program only seen in germline stem cells."
Ken Moberg PhD, Associate Professor, Cell Biology, Emory University School of Medicine
Researchers were then able to detect a physical interaction between Yorkie and Taiman, a protein critical in ecdysone response. The closest human protein relative to taiman is Amplified in Breast Cancer-1 or AIB1. This observation points to a possible connection between human AIB1 and the fruitfly Yorkie homologs Yap1 and Taz, states Moberg.
Research results are published in Developmental Cell.
In humans, the Hippo pathway is known to be involved in (1) forming embryonic stem cells, (2) suppressing cancerous growth, and also in (3) regenerative growth and (4) wound healing.
Ken Moberg PhD: "Since both groups of proteins are often over expressed in human cancers, we believe our findings have implications for the study of proliferative mechanisms in human cancer and possibly cancer stem cells."
Abstract Highlights
•The Drosophila EcR coactivator Tai binds the Hippo pathway coactivator Yki
•Tai modulates expression of classic Yki target genes in developing imaginal discs
•Tai’s role in Yki-driven hyperplasia is independent of classic Yki target genes
•Tai links hyperactive Yki to expression of germline mRNAs in disc cells
Summary
The Hippo pathway is a conserved signaling cascade that modulates tissue growth. Although its core elements are well defined, factors modulating Hippo transcriptional outputs remain elusive. Here we show that components of the steroid-responsive ecdysone (Ec) pathway modulate Hippo transcriptional effects in imaginal disc cells. The Ec receptor coactivator Taiman (Tai) interacts with the Hippo transcriptional coactivator Yorkie (Yki) and promotes expression of canonical Yki-responsive genes. Tai enhances Yki-driven growth, while Tai loss, or a form of Tai unable to bind Yki, suppresses Yki-driven tissue growth. This growth suppression is not correlated with impaired induction of canonical Hippo-responsive genes but with suppression of a distinct pro-growth program of Yki-induced/Tai-dependent genes, including the germline stem cell factors nanos and piwi. These data reveal Hippo/Ec pathway crosstalk in the form a Yki-Tai complex that collaboratively induces germline genes as part of a transcriptional program that is normally repressed in developing somatic epithelia.
Postdoctoral fellow Can Zhang, PhD and MD, PhD student Brian Robinson are co-first authors of the Developmental Cell paper. Collaborators at University of Massachusetts, Boston led by Alexey Veraksa, PhD contributed to the paper.
Return to top of page
|
