|
|
|
Home | Pregnancy Timeline | News Alerts |News Archive Jul 27, 2015

Nerve axon segments (BLUE) and the synapses that form along them (RED and GREEN) above.
Image Credit: Winnie Wefelmeyer. MRC Centre for Developmental Neurobiology |
|
|
|
|
|
Mind the gap!
In biology, stability is very important. From body temperature to blood pressure and sugar levels, our body ensures that all of our systems remain within reasonable limits, without damaging extremes. Brain neurons also stabilise electrical activity to avoid becoming either overexcited or not excited enough.
Overexcited brain neurons could lead to epilepsy. Those not excited enough could be nonfunctional. A new study from scientists at the Medical Research Council (MRC) Centre for Developmental Neurobiology at University College London in the United Kingdom, reveals how they found neurons contain their electrical stability.
The research is published in the journal PNAS,
At the centre of this stability is a short segment of the axon called the axon initial segment (or AIS). The AIS is where electrical activity is initiated — where all the information from synapses is collected to produce an electrical current. Modulating or altering the frequency of electromagnetic vibration, the AIS could have an important impact on a cell's excitability - its ability to limit or increase an electrical current.
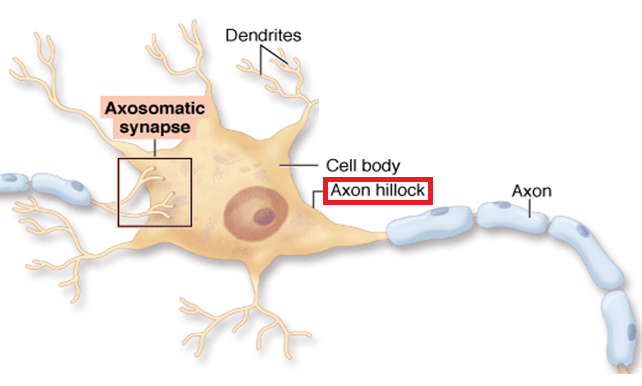
Previously, the lab of Juan Burrone PhD, Professor of Development Neurophysiology, the MRC Centre for Developmental Neurobiology, and senior author on the project, had uncovered that changes in the AIS structure could dampen the excitability of neurons and stabilize their electrical activity (Grubb & Burrone Nature 365:1070). But, not understood was what this process did to synapses formed along the AIS.
The study found increases in neuronal activity cause the AIS to move along the axon and away from the cell body, by about half the axon length. Evidence was found in slices of the hippocampus, a brain region important to learning and memory. The synapses, which control the AIS, had stayed in place - causing a mismatch with the AIS.
Computer models were then used to work out the functional outcomes of this arrangement revealing that synapses left behind, those that lie in the gap between the cell body and the AIS, are important in decreasing or dampening neuronal excitability. This allows neurons to remain functional even when under constant stimulation.
"The axon initial segment is like the vocal chord of the neuron: without it, the neuron would be unable to communicate. Changing the position of the AIS relative to the modulating synapses ensures what the neuron is saying stays meaningful."
Winnie Wefelmeyer PhD, ResearchAssociate, and lead author of the study, MRC Centre for Developmental Neurobiology, University College London, United Kingdom
Burrone: "Synapses are highly plastic. Both the number and strength of synaptic connections can vary dramatically in an activity-dependent manner, a property that is essential for information storage. However, plastic systems are inherently unstable and, if left unchecked, can result in networks drifting towards extreme levels of activity. As a result, neurons have developed ways to stabilize neuronal activity, generically known as homeostatic forms of plasticity.
"We propose that movement of the AIS along the axon may represent a novel mechanism for homeostatic plasticity, with neurons altering their subcellular organisation to fine-tune excitability in response to changes in ongoing activity levels. Together with the cell-wide changes in synaptic gain, neurons have devised multiple strategies to maintain their activity levels within a set range. Future experiments will study how these two forms of plasticity come together to modulate the input-output function of a neuron. More importantly, we believe that by understanding the mechanisms behind AIS relocation and synaptic homeostatic plasticity, we will uncover specific pathways that will provide possible targets for treating diseases where the activity of a network has become uncontrolled, such as in epilepsy."
Significance
Neurons integrate synaptic inputs at the axon initial segment (AIS), where the action potential is initiated. Recent findings have shown this structure to be highly plastic. Here, we focus on the axo-axonic synapses formed onto the AIS and show that although chronic increases in neuronal activity result in a distal relocation of the AIS, the synapses do not change position. Computational modeling suggests this spatial mismatch has critical functional consequences on neuronal output, allowing the cell to downregulate its own excitability and thus respond homeostatically to chronic stimulation.
Abstract
The axon initial segment (AIS) is a structure at the start of the axon with a high density of sodium and potassium channels that defines the site of action potential generation. It has recently been shown that this structure is plastic and can change its position along the axon, as well as its length, in a homeostatic manner. Chronic activity-deprivation paradigms in a chick auditory nucleus lead to a lengthening of the AIS and an increase in neuronal excitability. On the other hand, a long-term increase in activity in dissociated rat hippocampal neurons results in an outward movement of the AIS and a decrease in the cell’s excitability. Here, we investigated whether the AIS is capable of undergoing structural plasticity in rat hippocampal organotypic slices, which retain the diversity of neuronal cell types present at postnatal ages, including chandelier cells. These interneurons exclusively target the AIS of pyramidal neurons and form rows of presynaptic boutons along them. Stimulating individual CA1 pyramidal neurons that express channelrhodopsin-2 for 48 h leads to an outward shift of the AIS. Intriguingly, both the pre- and postsynaptic components of the axo-axonic synapses did not change position after AIS relocation. We used computational modeling to explore the functional consequences of this partial mismatch and found that it allows the GABAergic synapses to strongly oppose action potential generation, and thus downregulate pyramidal cell excitability. We propose that this spatial arrangement is the optimal configuration for a homeostatic response to long-term stimulation.
This study was supported by grants from the Wellcome Trust and the European Research Council.
The MRC Centre for Developmental Neurobiology is part of the Institute of Psychiatry, Psychology & Neuroscience at King's College London.
Return to top of page
|
