|
|
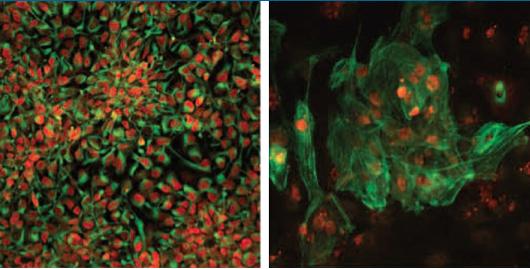
Salk researchers have generated disease-free stem cells from patients with mitochondrial disease
that can be converted into any cell type including (left) neuronal progenitors or (right) heart cells.
These newly generated cells could potentially be used for future transplantation into patients.
Image Credit: Salk Institute
|
|
|
|
|
|
Healthy brain, muscle, eye and heart cells would improve the lives of tens of thousands of people around the world with mitochondrial diseases. Now, researchers at the Salk Institute have gotten one step closer to making such cures a reality: they've turned patient's cells into healthy, mutation-free stem cells, with the ability to become any cell type.
"Right now, there are no cures for mitochondrial diseases. Very recently, we've developed ways to prevent these diseases, so it was natural to next ask how we could treat them."
Juan Carlos Izpisua Belmonte PhD, Professor, Gene Expression Laboratory, Salk Institute and senior author.
Mitochondrial diseases are caused by any of about 200 mutations that affect the genes of mitochondria, tiny powerhouses inside every cell of the body except red blood cells. They are responsible for creating more than 90% of the energy needed by the body to sustain life and support growth. When they fail, less and less energy is generated within the cell. Cell injury and sometimes cell death follow. If this process is repeated throughout the body, whole systems begin to fail and life is severely compromised. The disease primarily affects children, but adult onset is becoming more and more common.
Diseases of the mitochondria appear to cause the most damage to cells of the brain, heart, liver, kidney, endocrine and respiratory systems and the skeletal muscles. Existing therapies hope to ease the symptoms or slow the progression of a disease, but can't entirely cure.
In new work, Izpisua Belmonte and collaborators from around the world collected skin samples from patients with Leigh Syndrome or Mitochondrial Encephalomyopathy [brain and nervous system (encephalo-) and muscles (myopathy]. Most people with MELAS have a buildup of lactic acid in their bodies, a condition called lactic acidosis. Increased acidity of the blood can lead to vomiting, abdominal pain, extreme tiredness (fatigue), muscle weakness, and difficulty breathing. Less commonly, people with MELAS may experience involuntary muscle spasms (myoclonus), impaired muscle coordination (ataxia), hearing loss, heart and kidney problems, diabetes, and hormonal imbalances.
Leigh Syndrome is a severe neurological disorder that typically arises in the first year of life. This condition is characterized by progressive loss of mental and movement ability (psychomotor regression) and typically results in death within a couple of years, usually due to respiratory failure. A small number of individuals develop symptoms only in adulthood or have symptoms that worsen much more slowly.
Scientists began by using standard lab protocols to create pluripotent stem cells from a patien'ts skin cells, a process that resets the skin cells to their most basic state.
"During the process of stem cell generation, you spontaneously get different types of cloned cells," explains Alejandro Ocampo, a research associate in the Izpisua Belmonte lab and author on the paper. If the patient's cells had an initial mix of healthy and diseased mitochondria, healthy and diseased stem cells are generated. These stem cells can then be sorted by their healthy mitochondria. This new approach is described July 15, 2015 in Nature.
However, for some patients even this approach won't work as their cells don't contain enough — if any — healthy mitochondria. So the team came up with a second approach. Move the nucleus of the patient's skin cells, the cell nucleus contains most of our genes, into a donor egg cell with healthy mitochondria. Then, use the new egg cell to generate pluripotent stem cells. With this procedure the healthy mitochondria successfully took over cell function, generating healthy, genetically similar patient cells.
"In either case, we have healthy stem cells, and we know how to convert pluripotent stem cells into different cell types with the potential to give rise to every cell type in the body."
Jun Wu PhD, research associate in Belmonte's lab, Izpisua, and an author on the paper.
Now, researchers can use the healthy cells to generate heart, brain, muscle or eye cells from the mutation-free stem cells. But methods are still under development to grow those mutation-free stem cells to full maturity and function — before transplanting them back into patients.
As scientists have long struggled to understand why different organs and tissues are so differently affected by mitochondrial mutations, Izpisua Belmonte feels these two new methods will increase their ability to compare stem cells made from mitochondrial patients. Being able to coax each stem cell to develop into different cell types, an aspect of mitochondrial disease can now be studied in more detail.
Abstract
Pluripotency, the ability to generate any cell type of the body, is an evanescent attribute of embryonic cells. Transitory pluripotent cells can be captured at different time points during embryogenesis and maintained as embryonic stem cells or epiblast stem cells in culture. Since ontogenesis is a dynamic process in both space and time, it seems counterintuitive that these two temporal states represent the full spectrum of organismal pluripotency. Here we show that by modulating culture parameters, a stem-cell type with unique spatial characteristics and distinct molecular and functional features, designated as region-selective pluripotent stem cells (rsPSCs), can be efficiently obtained from mouse embryos and primate pluripotent stem cells, including humans. The ease of culturing and editing the genome of human rsPSCs offers advantages for regenerative medicine applications. The unique ability of human rsPSCs to generate post-implantation interspecies chimaeric embryos may facilitate our understanding of early human development and evolution.
Other researchers on the study were Li Ma of the Salk Institute; Hong Ma, Riffat Ahmed, Eunju Kang, Yeonmi Lee, Tomonari Hayama, Ying Li Crystal Van Dyken, Nuria Marti Gutierrez, Rebecca Tippner-Hedges, Amy Koski, Nargiz Mitalipov, Paula Amato, Don P. Wolf, and Shoukhrat Mitalipov of Oregon Health & Science University; Clifford D. L. Folmes, and Andre Terzic of the Mayo Clinic; Robert Morey, Sergio Mora-Castilla, and Louise C. Laurent of the University of California, San Diego; Joanna Poulton of the University of Oxford; and Xinjian Wang and Taosheng Huang of Cincinnati Children's Hospital.
The work was supported by the G. Harold and Leila Y. Mathers Charitable Foundation and the Leona M. and Harry B. Helmsley Charitable Trust.
About the Salk Institute for Biological Studies:
The Salk Institute for Biological Studies is one of the world's preeminent basic research institutions, where internationally renowned faculty probes fundamental life science questions in a unique, collaborative and creative environment. Focused both on discovery and on mentoring future generations of researchers, Salk scientists make groundbreaking contributions to our understanding of cancer, aging, Alzheimer's, diabetes and infectious diseases by studying neuroscience, genetics, cell and plant biology, and related disciplines. Faculty achievements have been recognized with numerous honors, including Nobel Prizes and memberships in the National Academy of Sciences. Founded in 1960 by polio vaccine pioneer Jonas Salk, MD, the Institute is an independent nonprofit organization and architectural landmark.
Introducing content sharing on nature.com
Macmillan Science and Education, the parent group of Nature Publishing Group, has launched an initiative that will make research articles on nature.com widely available to read and share. This was announced on 2 December 2014. Scientists have always shared their work, it is essential to advancing progress. Nature was established in 1869 to help scientists share, and to bring science to the public. In today's global, internet-enabled world, we think we can meet the needs of science and society better.
Nature Publishing Group (NPG) has now introduced experimental new functionality on nature.com and a beta set of principles and guidelines that will:
Enable subscribers to 49 journals on nature.com to share a read-only version of full text-subscription articles, to support collaboration. Reasonable sharing is encouraged for non-commercial, personal use.
Enable 100+ media outlets and blogs to link to a read-only version of full text-subscription articles
Return to top of page
|


