|
|
The father effect
How do environmental challenges get transmitted from a father to his grandchildren? If you have diabetes, or cancer or even heart problems, you might need to blame your dad's behavior or environment. Or even your grandfather's.
Why? Because, in recent years, scientists have shown that a father's life experiences with food, drugs, exposure to toxic products and even stress can affect the development and health not only of his children, but even of his grandchildren — before his offspring are even conceived.
Despite a decade of work, scientists still have much to learn about how environmental memories are transmitted over generations. McGill University of Montreal, Canada researchers along with their collaborators from the University of Basel, Basel, Switzerland and the University of Bristol, Bristol, UK, think they have now identified a key part in that molecular puzzle.
The team believes that proteins known as histones play a critical role in inheritance and are not simply the "spools" DNA loops around in order to compress into the cell.
This finding, just published in Science, has the potential to profoundly change our understanding of how we inherit. The researchers show there is something in addition to DNA that plays an important role in inheritance. Something that could determine whether a father's children and grandchildren will or will not be healthy.
To date, most of the research in this area, known as epigenetics, has focused on the methyl groups that attach to DNA and turn up or turn down the amount of protein expressed by specific genes on a DNA strand. The tighter the wrap of a methyl group, the less protein is available to be "read" by RNA and therefore less protein is expressed by the gene. Methyl groups act like a dimmer switch on a lamp cord.
Histones are distinct from DNA, acting as spools which DNA winds around. McGill researchers were curious if histones might play a role in transmitting heritable information as they are not only found in sperm, but transferred during fertilization to the newly made zygote.
To test that possibility, they created mice with altered biochemical data on their histones. During sperm cell formation, they measured samples to make sure the DNA "thread" looped around the altered histone "spools." They then looked for any affects these alterations might have on the offspring.
They discovered dire consequences to the offspring. The pups were prone to birth defects, abnormal skeletal formation, and death at birth. Most surprising, the effects could be seen for two generations.
"When we saw the decreased survivability across generations and the developmental abnormalities, we were really blown away. It was never thought that altering something outside of the DNA could affect inheritance," said Sarah Kimmins PhD, from McGill's Department of Animal Science, and one of the lead authors on the paper. Kimmins is also the Canada Research Chair in Epigenetics, Reproduction and Development.
Kimmins continued: "These findings are remarkable because they indicate that information other than DNA is involved in heritability. The study highlights the critical role that fathers play in the health of their children and even grand-children. Since chemical modifications on histones are susceptible to environmental exposures, the work opens new avenues of investigation for the possible prevention and treatment of diseases of various kinds, affecting health across generations."
"While there is substantial evidence that fathers can transmit diseases and adverse phenotypes to their children in the absence of genetic mutations, this is the first study that shows a feasible mechanism by which this can happen.
"This gives researchers confidence to pursue histone retention in the male germ cells as a mechanism of inheritance ... it also will serve as a reminder to fathers to be diligent protectors of their germline."
Marisa Bartolomei PhD, Proffessor, Dept. of Cell and Developmental Biology, Perelman School of Medicine, University of Pennsylvania
Abstract
A father’s lifetime experiences can be transmitted to his offspring to affect health and development. The mechanisms underlying paternal epigenetic transmission are unclear. Unlike somatic cells, there are few nucleosomes in sperm and their function in epigenetic inheritance is unknown. We generated transgenic mice in which overexpression of the histone H3 lysine 4 (H3K4) demethylase LSD1/KDM1A during spermatogenesis reduced H3K4 dimethylation in sperm. KDM1A overexpression in one generation severely impaired development and survivability of offspring. These defects persisted transgenerationally in the absence of KDM1A germ line expression and were associated with altered RNA profiles in sperm and offspring. We show that epigenetic inheritance of aberrant development can be initiated by histone demethylase activity in developing sperm, without changes to DNA methylation at CpG-rich regions.
Experts who have commented or are willing to be interviewed about the paper: John R. McCarrey, Robert and Helen Kleberg Distinguished Chair in Cellular & Molecular Biology, Department of Biology, University of Texas at San Antonio
Contacts: Prof. Sarah Kimmins, Dept. of Animal Science, McGill University (English interviews); Katherine Gombay, Media Relations Office, McGill University, 514-398-2189 http://www.mcgill.ca/newsroom/ ; http://twitter.com/McGillU
The research was funded by: Canadian Institutes of Health Research (CIHR), Genome Quebec, the Reseau de Reproduction Quebecois, Fonds de recherche Nature et technologies (FRQNT), Boehringer Ingelheim Fond, Swiss National Science Foundation and the Novartis Research Foundation.
Return to top of page
|
|
|
Oct 15, 2015 Fetal Timeline Maternal Timeline News News Archive
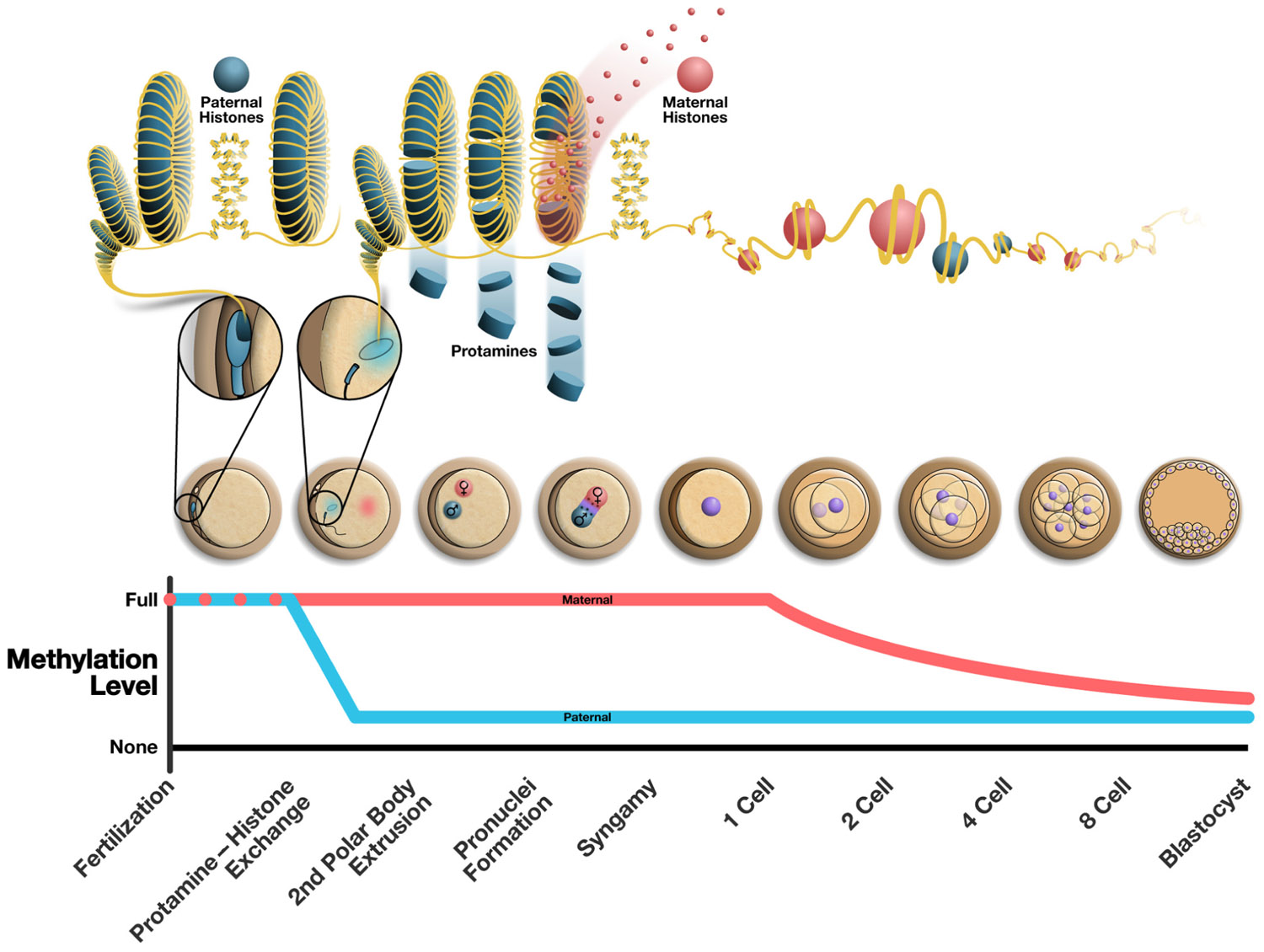
This diagram illustrates the current known breakdown of a mature sperm following fertilization.
Sperm chromatin is predominantly packaged by protamines [a type of protein]. Following
fertilization, the sperm pronucleus is remodeled and maternal histones replace protamines
loosening the complex of DNA, RNA and proteins found in the sperm head.
The middle panel illustrates various stages of early embryo development.
The bottom panel: the approximate chronology of major events in the early embryo
correlated to the embryo illustrations above..
Image Credit: Frontiers in Genetics
|
|
| |
|



