|
|
Cell signal strength turns on early stem cells
Stem cells work like handymen, repairing damaged tissues and renewing other tissues such as our skin. Scientists understand more about how stem cells work in adults, but less about how they work in an embryo.
Now, researchers at The Rockefeller University have identified the mechanism that instructs cells during development to become stem cells. Their work, published in the journal Cell, helps explain how communication between cells intervenes in the stem cell process, with hints for possibly treating skin cancer.
"While adult stem cells are increasingly well-characterized, we know little about their origins. Here, we show in skin progenitor stem cells - how hair follicles become specified as soon as cells divide and move downward to form an embryonic hair bud within the single-layer epidermis of an embryo. This timing is much earlier than we thought, giving us new insight into the establishment of these very special cells."
Elaine Fuchs PhD, Robin Chemers Neustein Laboratory of Mammalian Cell Biology and Development, The Rockefeller University, and a Howard Hughes Medical Institute investigator.
Which came first, the stem cell or the niche?
Clusters of stem cells receive signals from nearby cells instructing them to either stay a stem cell, or become a specific cell type. These instructive cells, called "niche" cells, maintain and support adult stem cell populations. Less understood, however, is how and when niche cells first appear in an embryo.
"Adult stem cells depend on niche cells for instructions on both how to become a stem cell, and what size stem cell population to maintain.
"The question is, does the niche appear first and call other cells over to become stem cells? Or is it the other way around? Could stem cells be appearing elsewhere first and then recruiting stem cells to the niche."
Tamara Ouspenskaia PhD, first author
Working with the mouse hair follicle, a region containing active stem cells, Fuchs and her colleagues investigated the first cell divisions occurring as a hair follicle forms.
Hair follicles begin as a small bud called a placode, which develops into a tissue of multiple layers made up of different cell types.
By labeling cells in the placode and tracking their progeny, scientists determined that with each division, one daughter cell stays put, while the other escapes into a different layer.
Experiments revealed that the escapee daughter cell becomes a stem cell. This finding is significant as it is the earliest point in stem cell development ever detected, indicating stem cells may exist before a niche is formed.
Cells must flee their original "niche" in order to become stem cells.
How cells become various cell types — whether a liver or skin cell for example — depends on numerous factors including the molecular signals they receive from other cells that help turn specific genes on and off.
Fuchs and her colleagues observed how cell signals differ between two daughter cells with each becoming unique from the other and located in different enviornments. The team wants is to eventually describe all the distinctive features of how molecular signals seal a cell's fate throughout embryogenesis, and so far have found an escaped daughter cell inhabits an area with low levels of WNT signalling. In contrast, stem cells respond more to the cell-cell communication of SHH (Sonic Hedgehog) and expand symmetrically.
This interplay of WNT and SHH can be seen in developing hair bud cells and the two unique cell types produced.
"These cells must leave home, must leave an environment with high WNT signaling, in order to become stem cells.
"We observed that Sonix Hedge Hog (SHH), actually comes from cells with a high WNT signal. In order for the escaped cell to become a stem cell, it must receive an SHH signal from its sister cell saying: 'You are the stem cell!"
Tamara Ouspenskaia PhD
Researchers believe that antagonism between WNT and SHH signaling may help control the number of stem cells produced during embryo development.
Fuchs: "This newly identified signal crosstalk provides insight into why these two signals have such a profound impact on skin cancers, where the numbers of tissue-propagating stem cells are excessive. The work paves the way for future research into the fascinating and clinically important relationship between tumor-propagation and normal stem cells."
Abstract Highlights
•Asymmetric divisions within hair buds differentially partition WNT and SHH signaling
•This partitioning couples asymmetric cell division to asymmetric cell fate
•WNTlo daughters that respond to, but do not produce, SHH expand symmetrically
•WNThi SHH-producing cells ultimately cease producing stem cell daughters
Summary
Adult stem cell (SC) maintenance and differentiation are known to depend on signals received from the niche. Here, however, we demonstrate a mechanism for SC specification and regulation that is niche independent. Using immunofluorescence, live imaging, genetics, cell-cycle analyses, in utero lentiviral transduction, and lineage-tracing, we show that in developing hair buds, SCs are born from asymmetric divisions that differentially display WNT and SHH signaling. Displaced WNTlo suprabasal daughters become SCs that respond to paracrine SHH and symmetrically expand. By contrast, basal daughters remain WNThi. They express but do not respond to SHH and hence maintain slow-cycling, asymmetric divisions. Over time, they become short-lived progenitors, generating differentiating daughters rather than SCs. Thus, in contrast to an established niche that harbors a fixed SC pool whose expelled progeny differentiate, asymmetric divisions first specify and displace early SCs into an environment conducive to expansion and later restrict their numbers by switching asymmetric fates.
Return to top of page
|
|
|
Jan 18, 2016 Fetal Timeline Maternal Timeline News News Archive
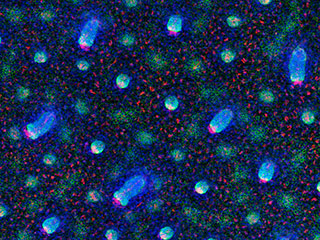
Researchers tracked cell divisions that occur as hair follicles form in mice, to determine
where stem cells first emerge. Above, developing hair follicles are shown at various stages.
Image Credit: Fuchs laboratory, Rockefeller University
|
|
|
|



